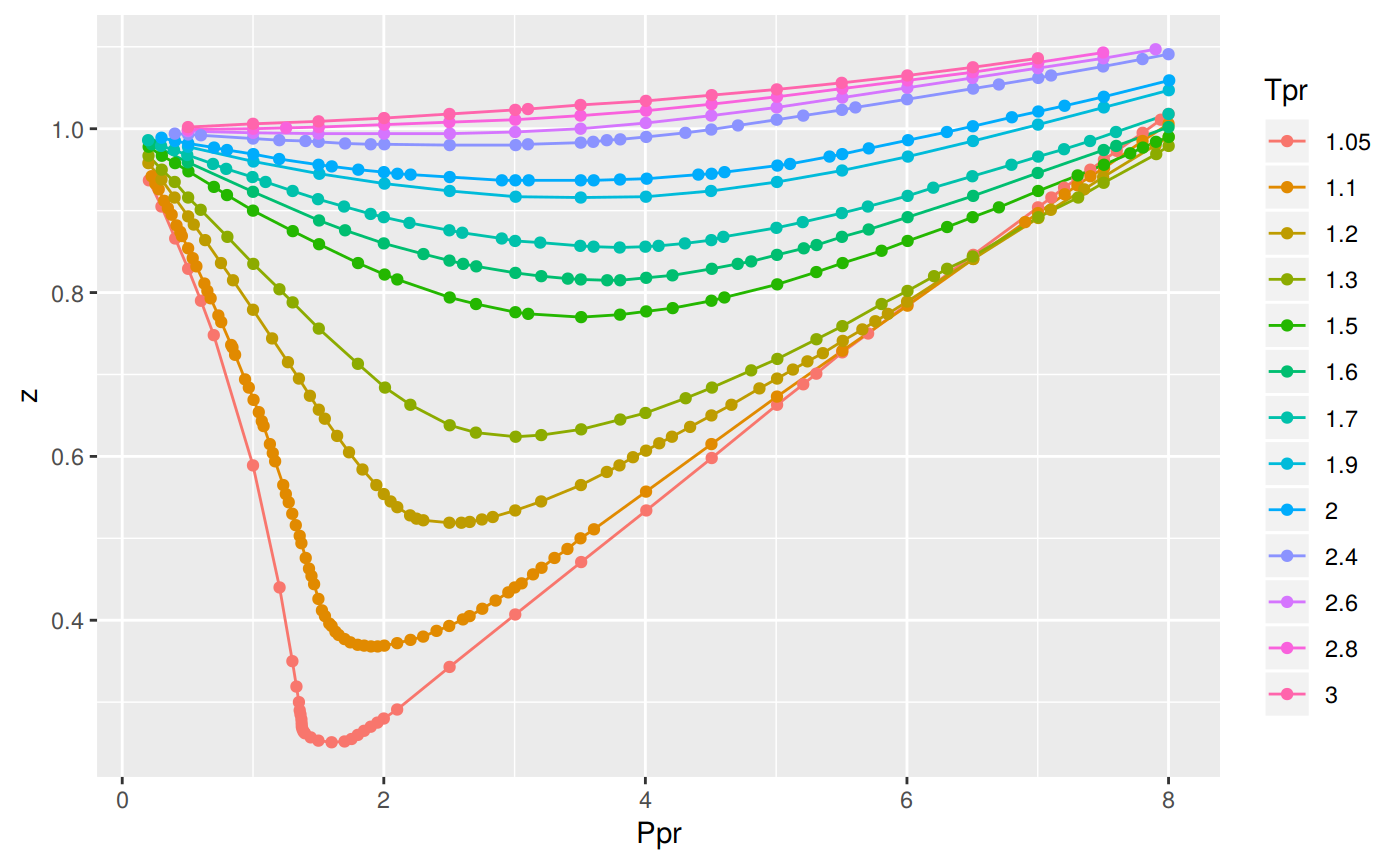
Write the expression for the compressibility factor (Z) for one mole of a gas. Write the value of Z for an
By A Mystery Man Writer
Description

daniloteste by Danilo Matteus - Issuu
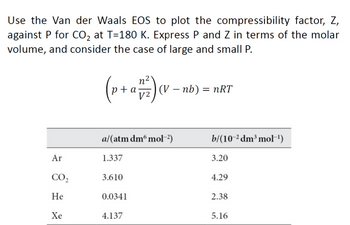
Answered: Use the Van der Waals EOS to plot the…

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

SOLVED: Derive the mathematical expression expressing the compressibility factor Z of a real gas depending on the reduced variables; Explain in detail how the volume of the actual gas at a given

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

A predictive PC-SAFT EOS based on COSMO for pharmaceutical compounds

For H(2) gas, the compressibility factor,Z = PV //n RT is

Answered: Compression factor of a gas with van…

Part 4. Thermodynamics of Gases - W.H. Freeman
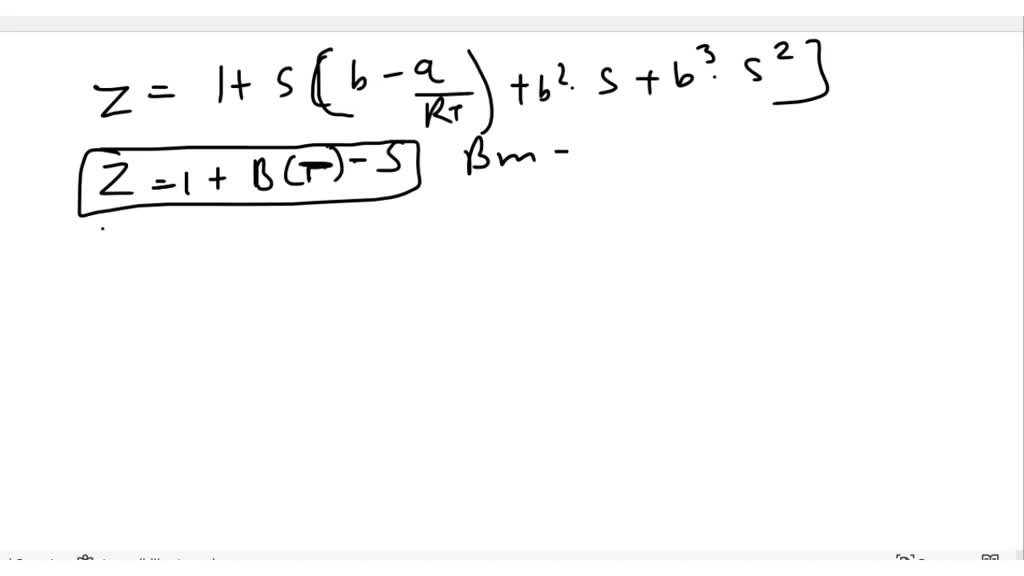
SOLVED: Derive the mathematical expression expressing the compressibility factor Z of a real gas depending on the reduced variables; Explain in detail how the volume of the actual gas at a given
from
per adult (price varies by group size)