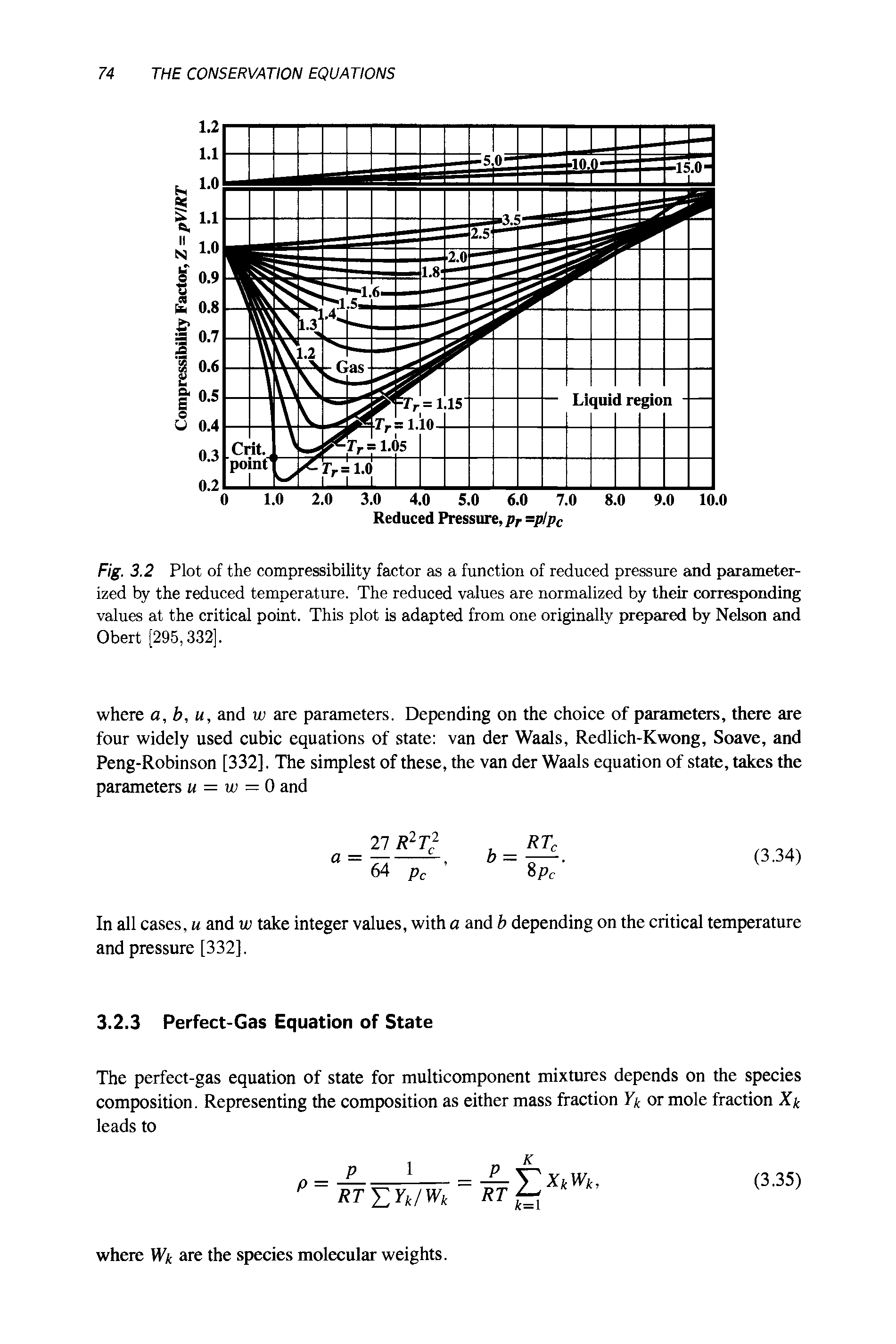
For $CO$, isotherm is of the type as shown. Near the point compressibility factor $Z$ is?\n \n \n \n \n 1.$\\left( {1 + \\dfrac{b}{V}} \\right)$ 2.$\\left( {1 - \\dfrac{b}{V}} \\right)$3.$\\left( {1 + \\
By A Mystery Man Writer
Description
For $CO$, isotherm is of the type as shown. Near the point compressibility factor $Z$ is?\n \n \n \n \n 1.$\\left( {1 + \\dfrac{b}{V}} \\right)$ 2.$\\left( {1 - \\dfrac{b}{V}} \\right)$3.$\\left( {1 + \\dfrac{a}{{RTV}}} \\right)$4.$\\lef

Solved () tor α = (*), P The coefficient of isothermal

⏩SOLVED:For liquid water the isothermal compressibility is given by:…
For CO, isotherm is of the type as shown.Near the pointA , compressibility factor Z is
52. For CO, isotherm is of the as shown. Near the point A, compressibility factor Z is CO Ideal

Thermodynamics - Test 1 Problem 5 - Ideal Gas Equation with Compressibility Factor

Compressibility factor - Wikipedia
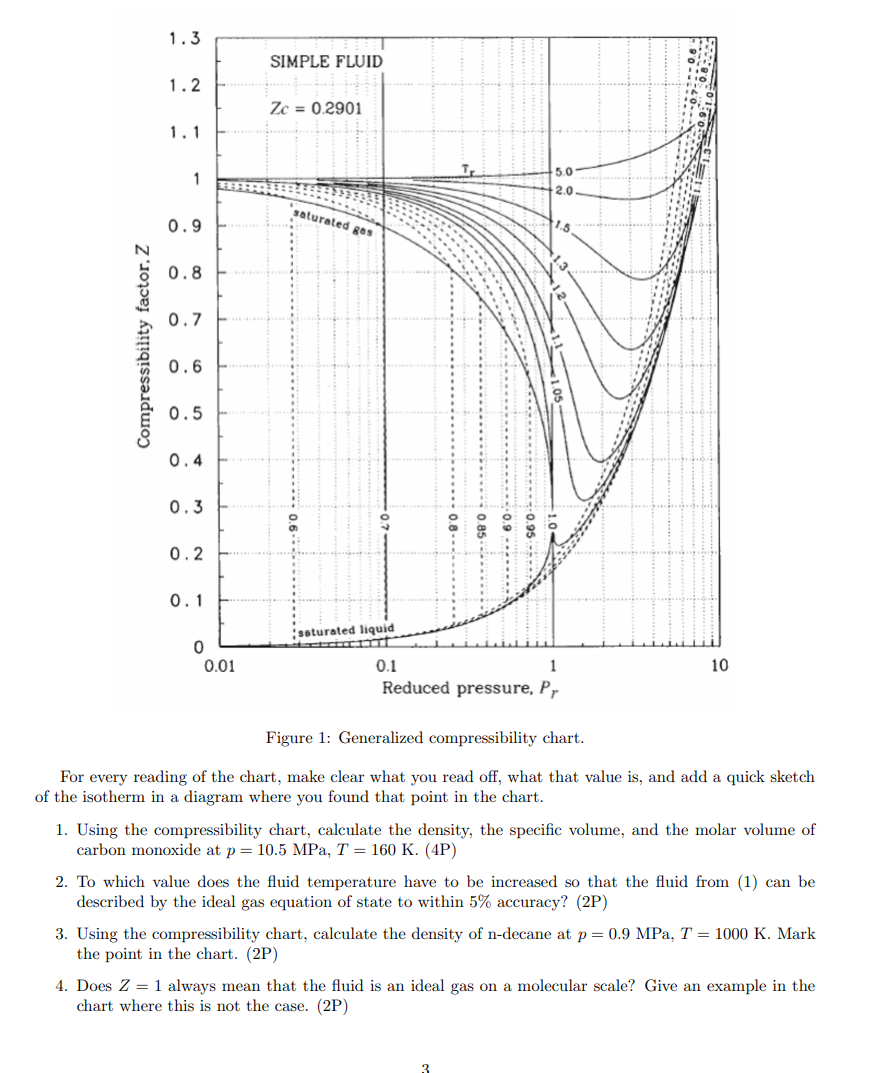
Solved 1.3 SIMPLE FLUID 1.2 Zc = 0.2901 1.1 1 5.0 2.0

The value of compressibility factor at the critical state the gas matches with the `Z_(c )` is

Thermodynamics - Test 1 Problem 5 - Ideal Gas Equation with Compressibility Factor
from
per adult (price varies by group size)