At low pressure, the van der waal's equation is written as (P+ a/V
By A Mystery Man Writer
Description
At low pressure, the van der waal's equation is written as (P+ a/V^2)V=RT . Then compressibility factor is then equal to :
At low pressure- the van der waal-s equation is written as -P- a-V-2-V-RT - Then compressibility factor is then equal to
At low pressure- the van der waal-s equation is written as -P- a-V-2-V-RT - Then compressibility factor is then equal to

At low pressures (for 1 mole), the van der Waal's equation is

Help with Van der Waals' equation : r/chemistry

The equation of state for real gas is given by (P+a/V2)(V b)=RT

Van der Waals Equation: Definition, Derivations, and Examples

answer the following 84 At a moderate pressure, the van der Waals
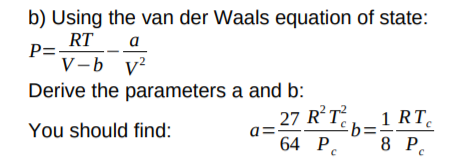
Answered: b) Using the van der Waals equation of…

Van Der Waals Equation - an overview
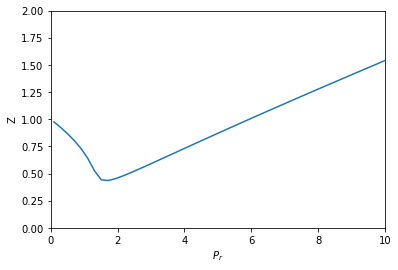
Compressibility factor variation from the van der Waals equation

Why do we use the ideal gas equation when instead van der Waals
from
per adult (price varies by group size)












