Equation of state (excess compressibility factor, Z À1 ¼ PV/(NkT) À1
By A Mystery Man Writer
Description

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

PDF) CALCULATION OF Z FACTORS FOR NATURAL GASES USING EQUATIONS OF STATE GAS TECHNOLOGY Calculation of Z Factors For Natural Gases Using Equations of State
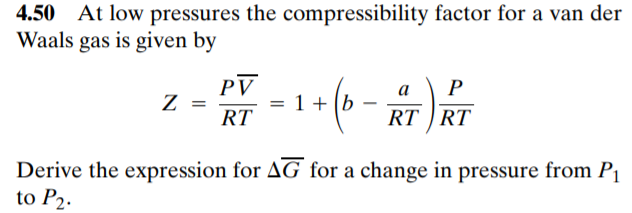
Solved 4.50 At low pressures the compressibility factor for

Solved 2. (20 points) At low pressures, the compressibility

Chapter 1 Overview of Chemical Reaction Engineering - CNTQ

Compressibility factor `Z=(PV)/(RT)`. Considering ideal gas, real gas, and gases at critical

Equation of state (excess compressibility factor, Z À1 ¼ PV/(NkT) À1
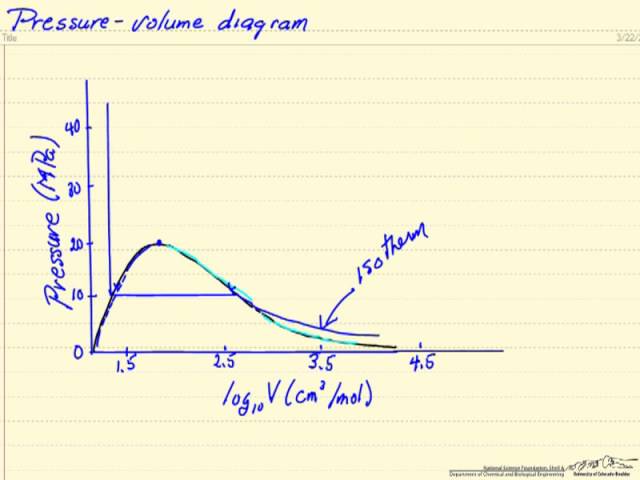
Compressibility Factor (Z-Factor) Equation of State

The compressibility factor of a gas is defined as Z=PV/nRT. The compressibility factor of an ideal gas is:1-1zeroinfinite
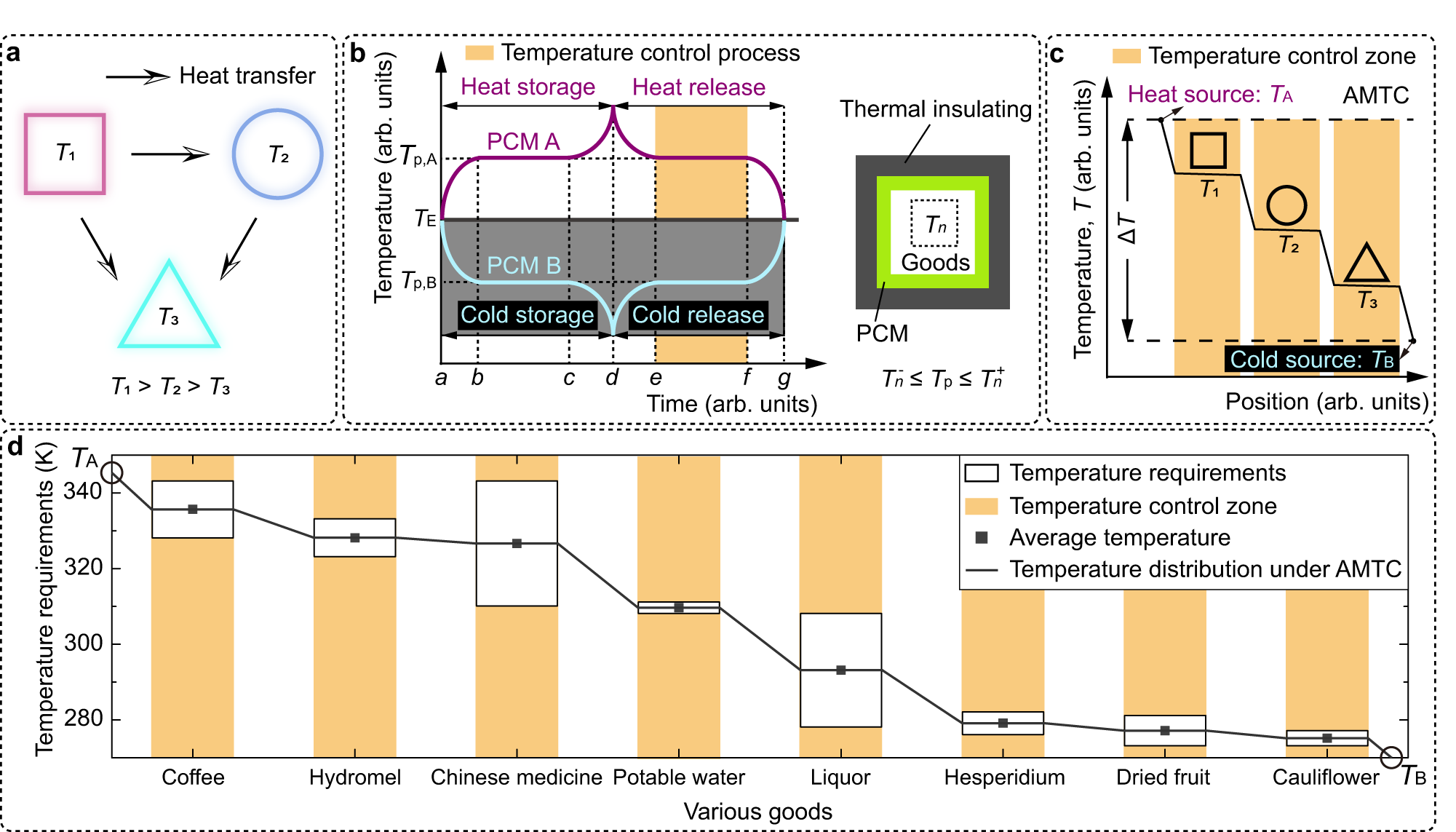
Adaptive multi-temperature control for transport and storage containers enabled by phase-change materials

Review of the Decomposition of Ammonia to Generate Hydrogen

Polymers, Free Full-Text

Equation of state (excess compressibility factor, Z À1 ¼ PV/(NkT) À1
from
per adult (price varies by group size)