total number of atoms in 44 g of Carbon dioxide is ?
By A Mystery Man Writer
Description
Total number of atoms in 44 g of Carbon dioxide is
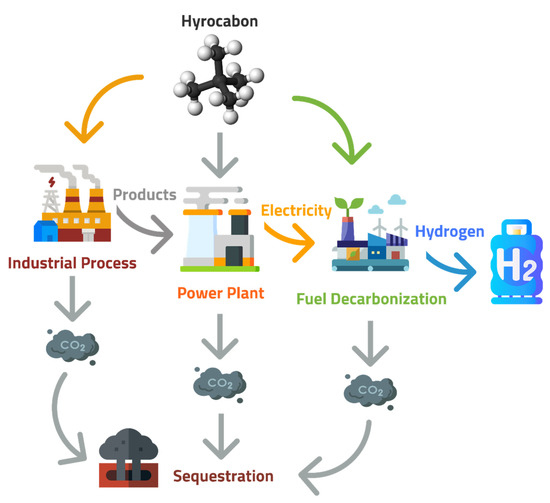
ChemEngineering, Free Full-Text

The number of oxygen atoms in 4.4 g of CO(2) is approximately

SOLVED: How many grams of carbon dioxide, CO2, (MW = 44.01 g/mol) occupy a volume of 15.3 L at STP? Group of answer choices 0.638 g 0.348 g 0.683 g 30.1 g 7.79 g

SOLVED: calculate the total number of atoms present in 44 gram of carbon dioxide

How many atoms are present in 44 gm of CO2? - Quora

SOLVED: calculate the total number of atoms present in 44 gram of carbon dioxide

Understanding carbon dioxide activation and carbon–carbon coupling over nickel

Solved 1) How many O atoms are in 4.39 g of CO2? Molar Mass

How many carbon atoms are present in 0.44 g carbon dioxide? (A) 10-2 (B) 6.022 x 1021 (C) 6.022 x 1025 (D) 2 x 10-2 min 0.44 9 cm3 6.02 103
from
per adult (price varies by group size)







