What is the mass of glucose required to produce 44g of C{O_{2'}} on complete combustion?30g45g60g22g
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:what is the mass of glucose required to produce 44g of co2 on complete
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g
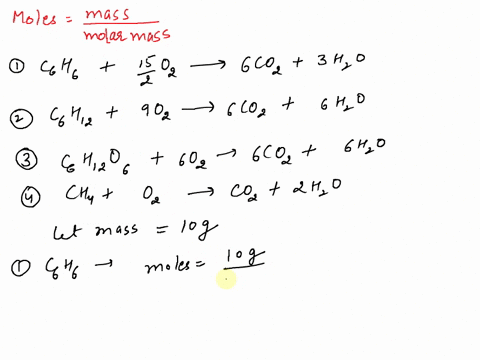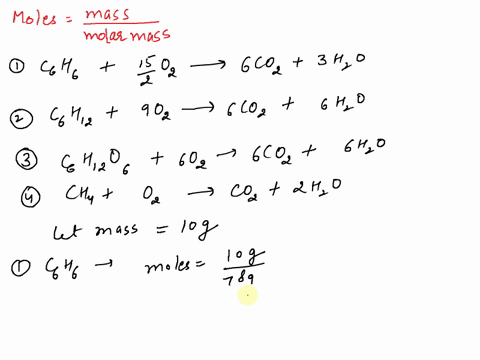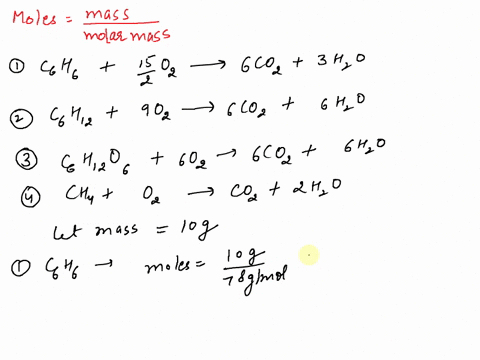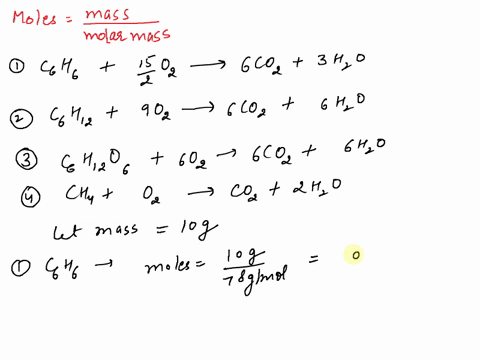
SOLVED: If equal masses of the following compounds undergo complete combustion, which will yield the greatest mass of CO2? 1. Benzene, C6H6 2. Cyclohexane, C6H12 3. Glucose, C6H12O6 4. Methane, CH4

Competitive Exams (Level-1) Some Basic Concepts What is the mass of glucose required to produce 44 g of Co, on complete combustion? (2) 459 (1) 30 g (4) 229 (3) 60 g

Solved A sample of a compound that contains only the

Unit 1 Stoichiometry Flashcards
Mass of glucose required to produce 44g of co2 on complete oxidation?
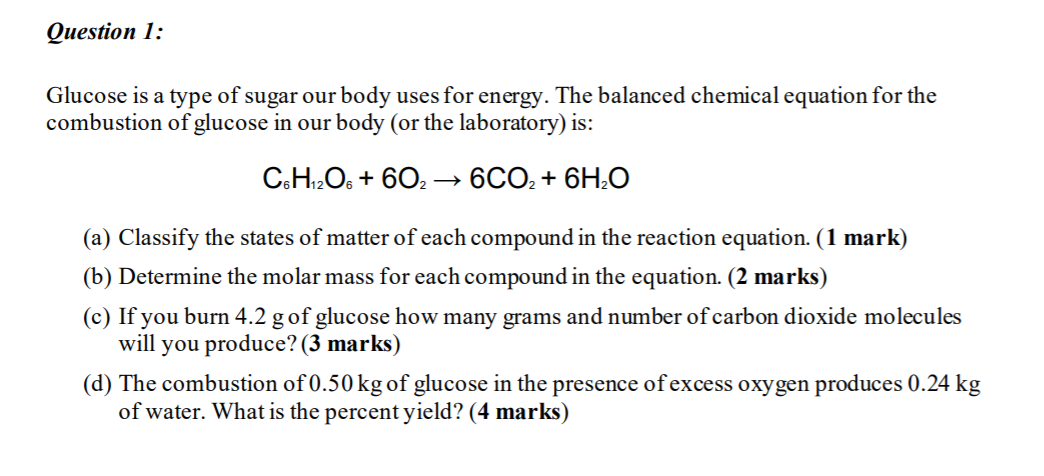
Solved Question 1: Glucose is a type of sugar our body uses

Solved 1. The combustion of sugar produces carbon dioxide
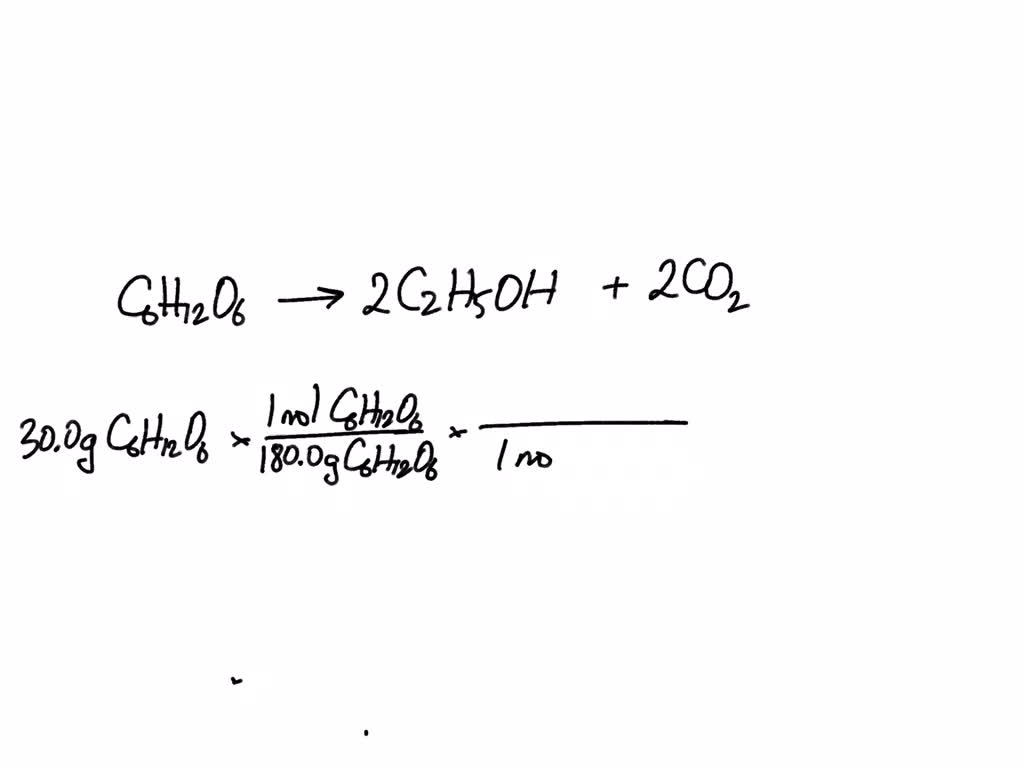
SOLVED: (ii) Calculate the maximum mass of ethanol that could be obtained from 30.0g of glucose
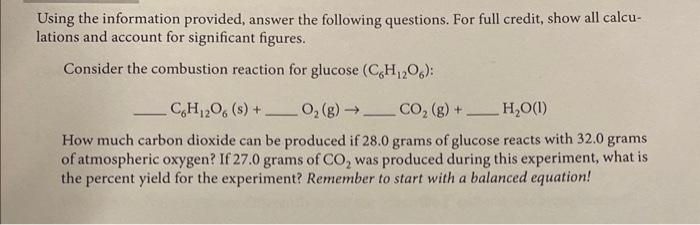
Solved Using the information provided, answer the following

Solved Fructose is a sugar commonly found in fruit. A sample
from
per adult (price varies by group size)







