Solved What is the equilibrium constant (Kp) at 45 °C for
By A Mystery Man Writer
Description
Answer to Solved What is the equilibrium constant (Kp) at 45 °C for
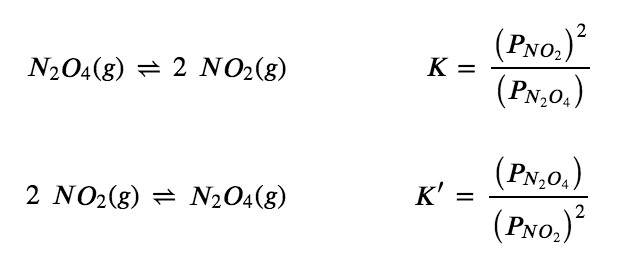
4.2 – The Equilibrium Constant & Reaction Quotient – General Chemistry for Gee-Gees
Equilibrium Constant (Kc, Kp) - Definition, Applications, Formula

The equilibrium constant (KP) of the reactions N2O4hArr 2NO2 was found
search-static.byjusweb.com/question-images/aakash_

16.41a Calculate the equilibrium constant at 25 °C for O2(g) + 2F2(g) → 2OF2(g) ΔG° = −9.2 kJ
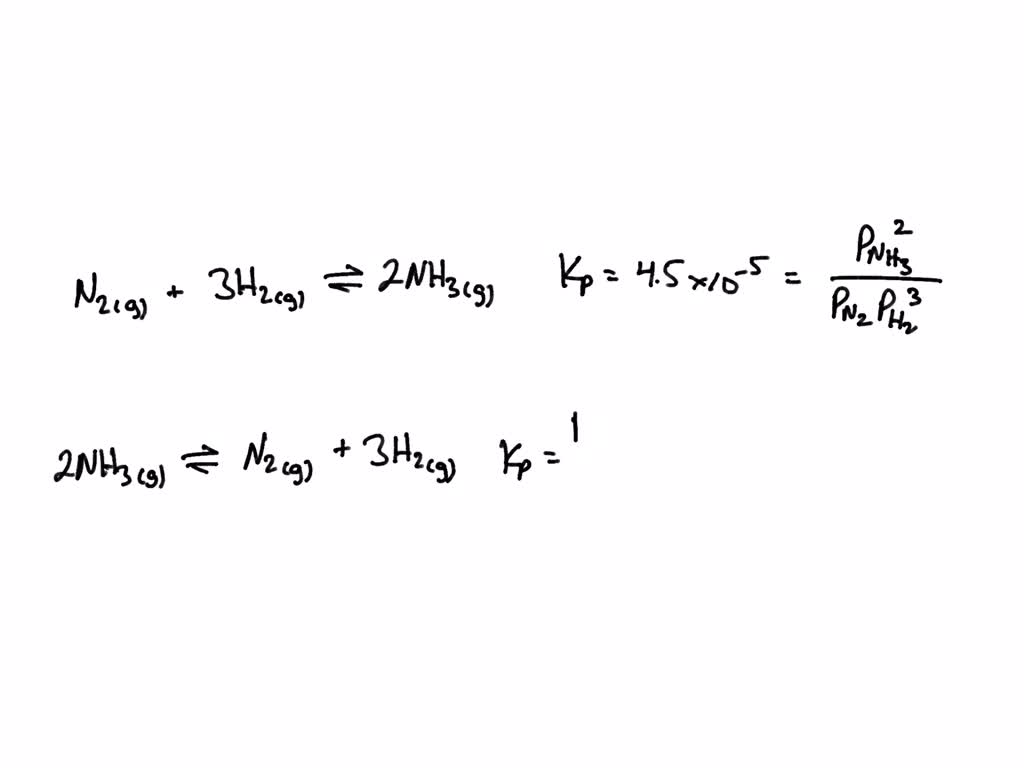
SOLVED: The value of the equilibrium constant Kp for the formation of ammonia N2(g) + 3H2(g) ⇌ 2NH3(g) is 4.5 x 10^5 at 450 °C. What is the value of Kp at

Solved the equilibrium constant, K, is 6.3x10 s at l500K for

The equilibrium constant K(p) for the thermal dissociation of PCl(5) a

Consider the reaction: A(g) ⇌ B(g) + C(g) Find the equilibrium co

Consider the reaction: A(g) ⇌ B(g) + C(g) Find the equilibrium co

i.ytimg.com/vi/4dKCx2crbG8/maxresdefault.jpg

How to Calculate the Equilibrium Constant, K
from
per adult (price varies by group size)