What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
By A Mystery Man Writer
Description
What is the value of compressibility factor in terms of vander waal cons†an t at different conditions of pressure and volume?Why is Z>1 for H2 and He gas
What is the value of compressibility factor in terms of vander waal cons-an t at different conditions of pressure and volume-Why is Z-1 for H2 and He gas
What is the value of compressibility factor in terms of vander waal cons-an t at different conditions of pressure and volume-Why is Z-1 for H2 and He gas

PDF) Measurement and Prediction Method of Compressibility Factor at High Temperature and High Pressure

Physical Chemistry The Compression Factor (Z) [w/1 example]

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application
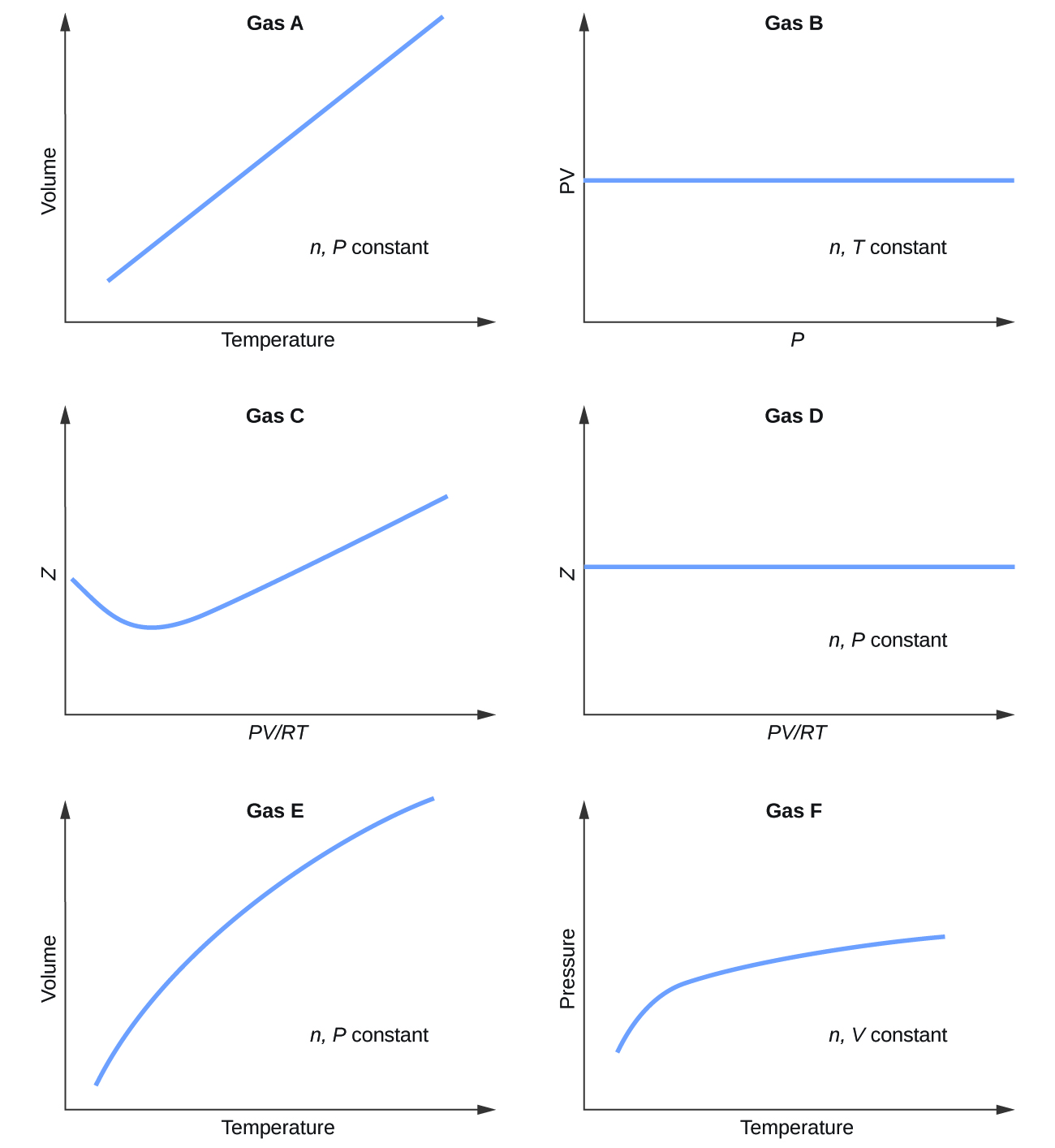
Non-Ideal Gas Behavior – Chemistry

Compressibility factor - Wikipedia

16.4: The Law of Corresponding States - Chemistry LibreTexts
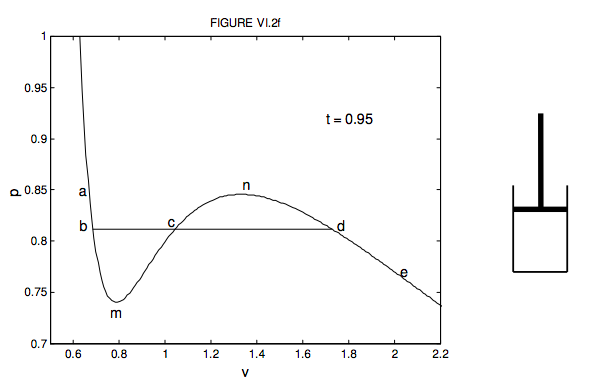
6.3: Van der Waals and Other Gases - Physics LibreTexts

SOLVED: Consider the Van der Waals equation of state (P + a/V^2)(V - b) = RT (a) Use this equation of state together with the table of a and b values below

Solved Problem 2. ( 30 points) The ideal gas law can

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

At a high pressure, the compressibility factor (Z) of a real gas is us

Compressibility factor vs. number of cycles for a typical isobar.
from
per adult (price varies by group size)





