Semi-synthetic transformations of ajugasterone C (1), poststerone
By A Mystery Man Writer
Description

Ecdysteroids as defensive chemicals - ScienceDirect

Máté VÁGVÖLGYI, Research Fellow, Ph.D., University of Szeged, Szeged, Institute of Pharmacognosy

Semi-synthetic transformations of ajugasterone C (1), poststerone (8)
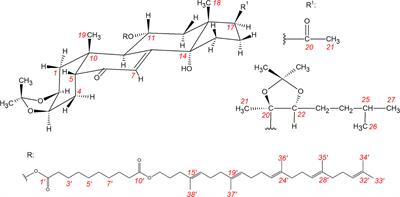
Frontiers Squalenoylated Nanoparticle Pro-Drugs of Adjuvant Antitumor 11α-Hydroxyecdysteroid 2,3-Acetonides Act as Cytoprotective Agents Against Doxorubicin and Paclitaxel
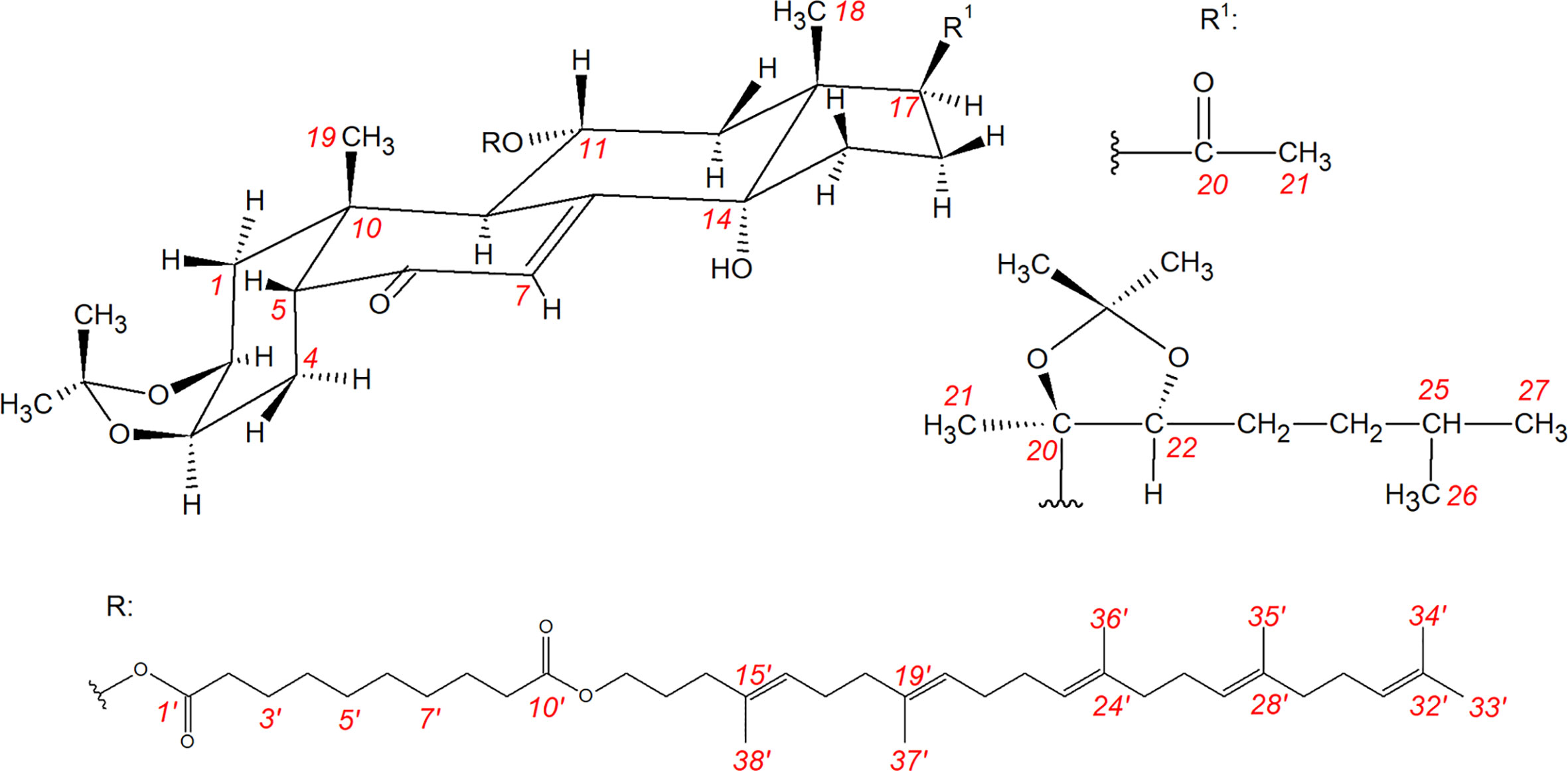
Frontiers Squalenoylated Nanoparticle Pro-Drugs of Adjuvant Antitumor 11α-Hydroxyecdysteroid 2,3-Acetonides Act as Cytoprotective Agents Against Doxorubicin and Paclitaxel

Identification on Daily Mortality Cause of Larvae in Treatment A1 (0 mg

PDF) Backstabbing P-gp: Side-Chain Cleaved Ecdysteroid 2,3-Dioxolanes Hyper-Sensitize MDR Cancer Cells to Doxorubicin without Efflux Inhibition

Tamás Gáti's research works Semmelweis University, Budapest (SOTE) and other places
Effect on proliferation (A) and viability (B) in the A431 cancer cell

Ecdysteroids as defensive chemicals - ScienceDirect

8 normal web of Trichonephila clavipes9 cocoon of H. bicolor attached

Ecdysteroids: isolation, chemical transformations, and biological activity

Microbiology Research, Free Full-Text
from
per adult (price varies by group size)







