Welcome to Chem Zipper.com: The compressibility factor for 1 mole of a van der Waals gas at 0oC and 100 atm pressure is found to be 0.5. Assuming that the volume of
By A Mystery Man Writer
Description

1148 questions with answers in GAS

The compression factor (compressibility factor) one mole of a van der Waals' gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is

Solved We begin by showing that the compressibility factor

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

Solved (Triple-Play Bonus) For a certain gas, the
How do Van der Waals constants a and b depend on temperature, pressure and volume? - Quora
The compression factor (compressibility factor) for one mole of a van der Waals' gas at 0ºC and 100 atm pressure is - Sarthaks eConnect
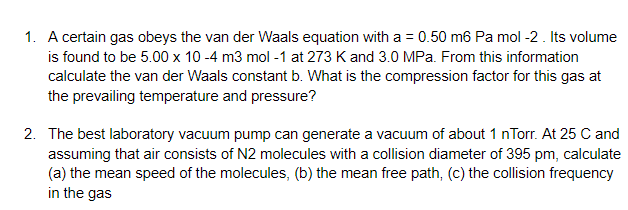
Solved A certain gas obeys the van der Waals equation with a

Water movement and its utilization by plants under the deep water storage in soil by using off-season irrigation

The compression factor (compressibility factor) one mole of a van der Waals'gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is negligible

Solved 1C.9(b) A certain gas obeys the van der Waals

The compression factor (compressibility factor) one mole of a van der Waals'gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is negligible

The compression factor (compressibility factor) for 1 mol of a van der

Kirkwood–Buff-Derived Force Field for Peptides and Proteins: Applications of KBFF20

Solved 2. Fugacity for a van-der-Waals gas Let's get a feel
from
per adult (price varies by group size)






