Derive an expression for the compression factor of a gas tha
By A Mystery Man Writer
Description
Energies, Free Full-Text

Solved 1, A gas at 250 K and 1 atm has a molar volume 5%

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson
PPT - ERT 108 Physical Chemistry INTRODUCTION-Part 2 PowerPoint Presentation - ID:2630974

SOLVED: The Berthelot equation of state is given by: p = nRT / (V - nb) The total differential of the compression factor Z is given by: dZ = MdT + NdV (
Answer in General Chemistry for Carl #275533

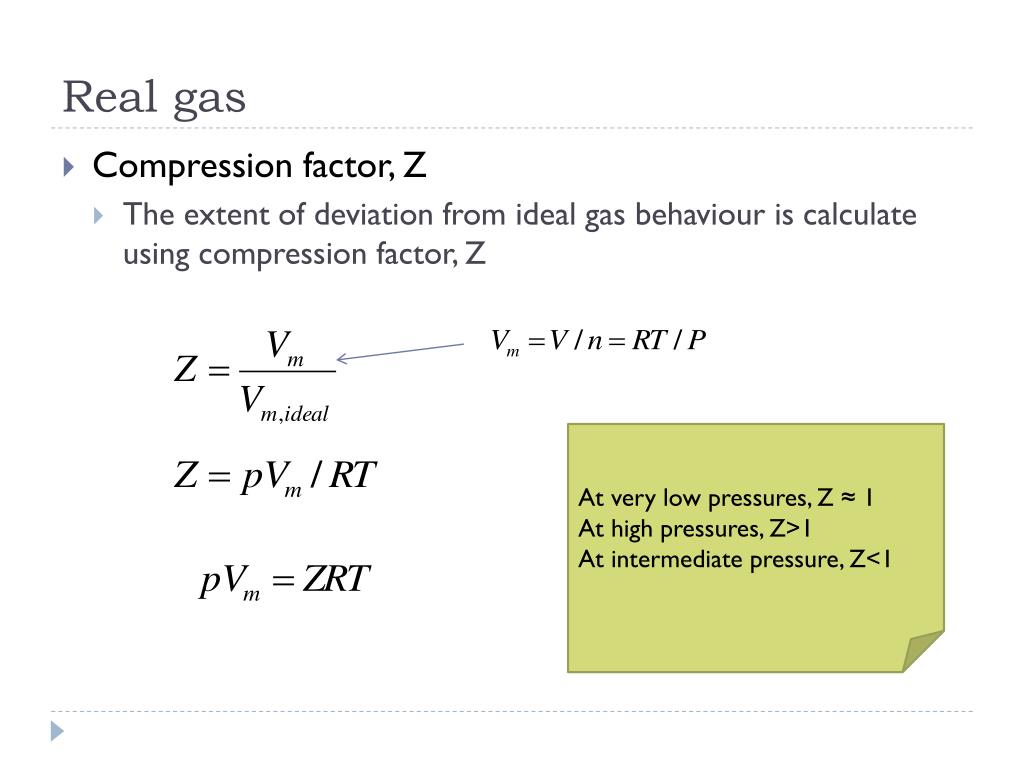
Compressibility factor - Wikipedia
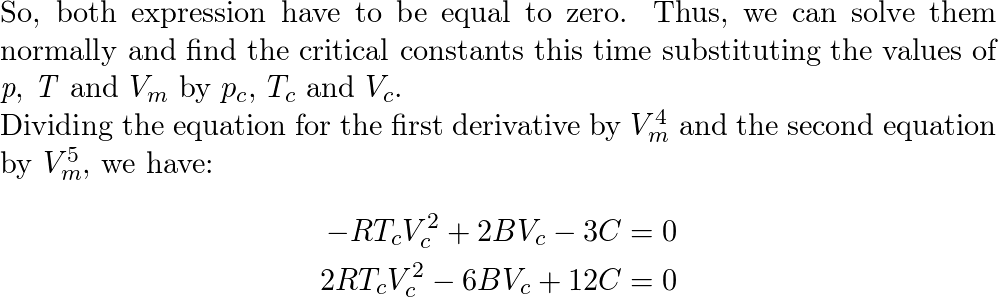
A scientist proposed the following equation of state $p=
ra

1. A gas at 250 K and I atn has a molar volume 5%

Answered: α = alpha A possible equation of state…

Van der Waals equation: van der Walls EOS, [Pr*3/Vr^2] [3Vr-1] =

SOLVED: State (i) the ideal gas equation (ii) the van der Waal's equation of state. Derive an expression for the compression factor; Z of a van der Waals gas in terms of
from
per adult (price varies by group size)