Compressibility factor Z = PV / nRT is plotted against pressure as
By A Mystery Man Writer
Description
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2

Real gases
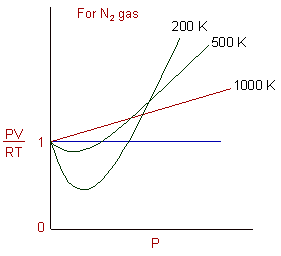
REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR

A real gas M behaves almost like an ideal gas. Graph 1 is obtained by plotting volume, V against temperature, T for x mol of gas M at pressure, P_1. a. Suggest

The given graph represent the variations of Z (compressibility factor (Z)=dfrac {pV}{nRT}) versus P, three real gases A, B and C. Identify the only incorrect statement.For the gas B, b=0 and its

Compressibility factor - Wikipedia

Van der Waals equation - Wikipedia

What is compressibility factor? What is its value for ideal gas

Compressibility Factor Charts - Wolfram Demonstrations Project

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure
Essential Pharma Documents: 1205: Properties of Gases
from
per adult (price varies by group size)