Gas compressibility factor Z: Ideal gas vs Real gas
By A Mystery Man Writer
Description
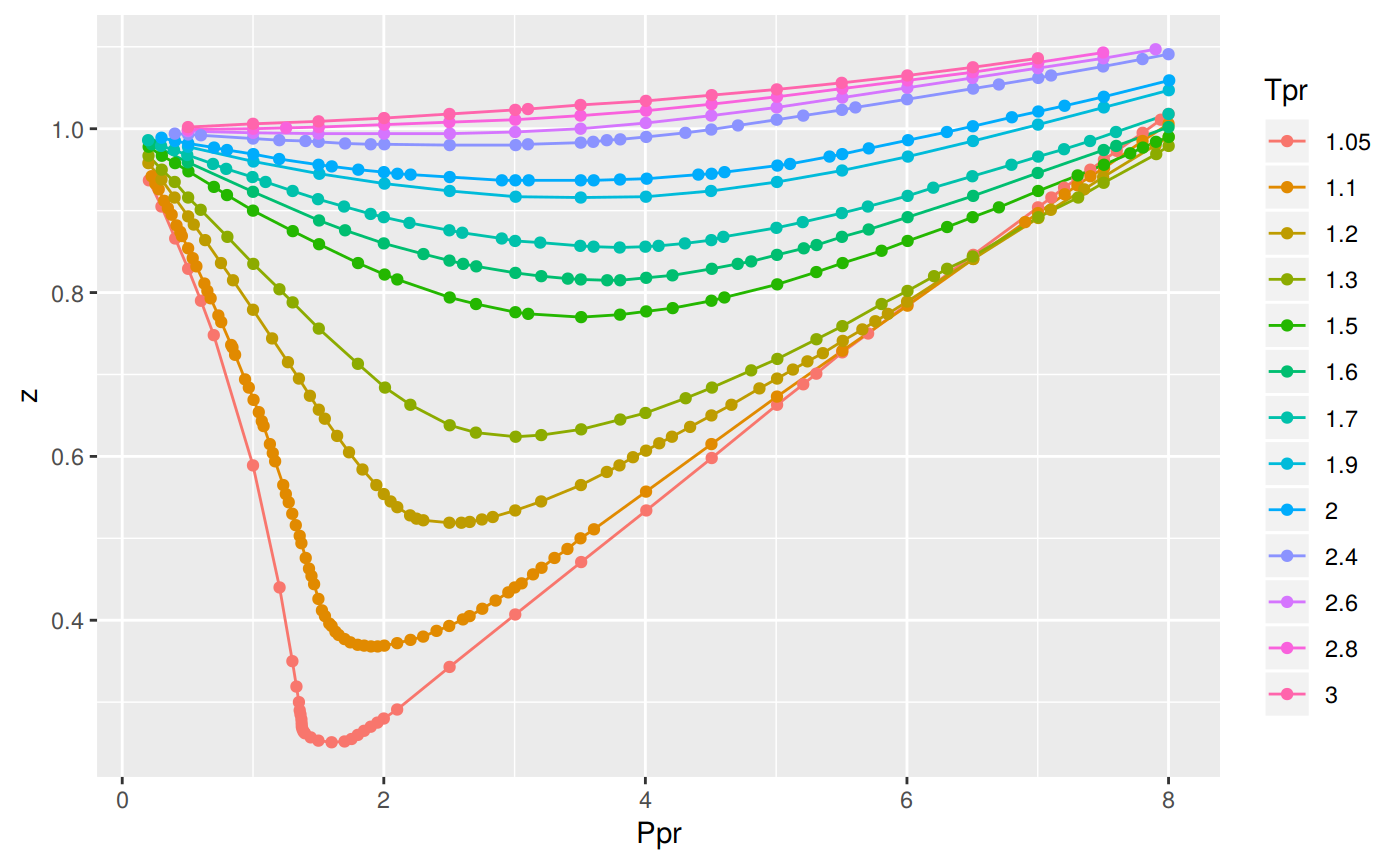
Gas compressibility factor, Z, and Gas compressibility are not the same. Gas compressibility factor Z is the ratio of the gas volume at a given temperature and pressure to the volume the gas would occupy if it were an ideal gas at the same temperature and pressure.

Real gas z-Factor chart [2] Download Scientific Diagram

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

gas laws - Graph of compressibility factor vs pressure when real gas is assigned Z=1 - Chemistry Stack Exchange

Acidizing 1. Types of Acid Treatments

Real Gases and Compressibility Factor

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Centrifugal Compressor Surging Causes

Compressibility factor for real gases
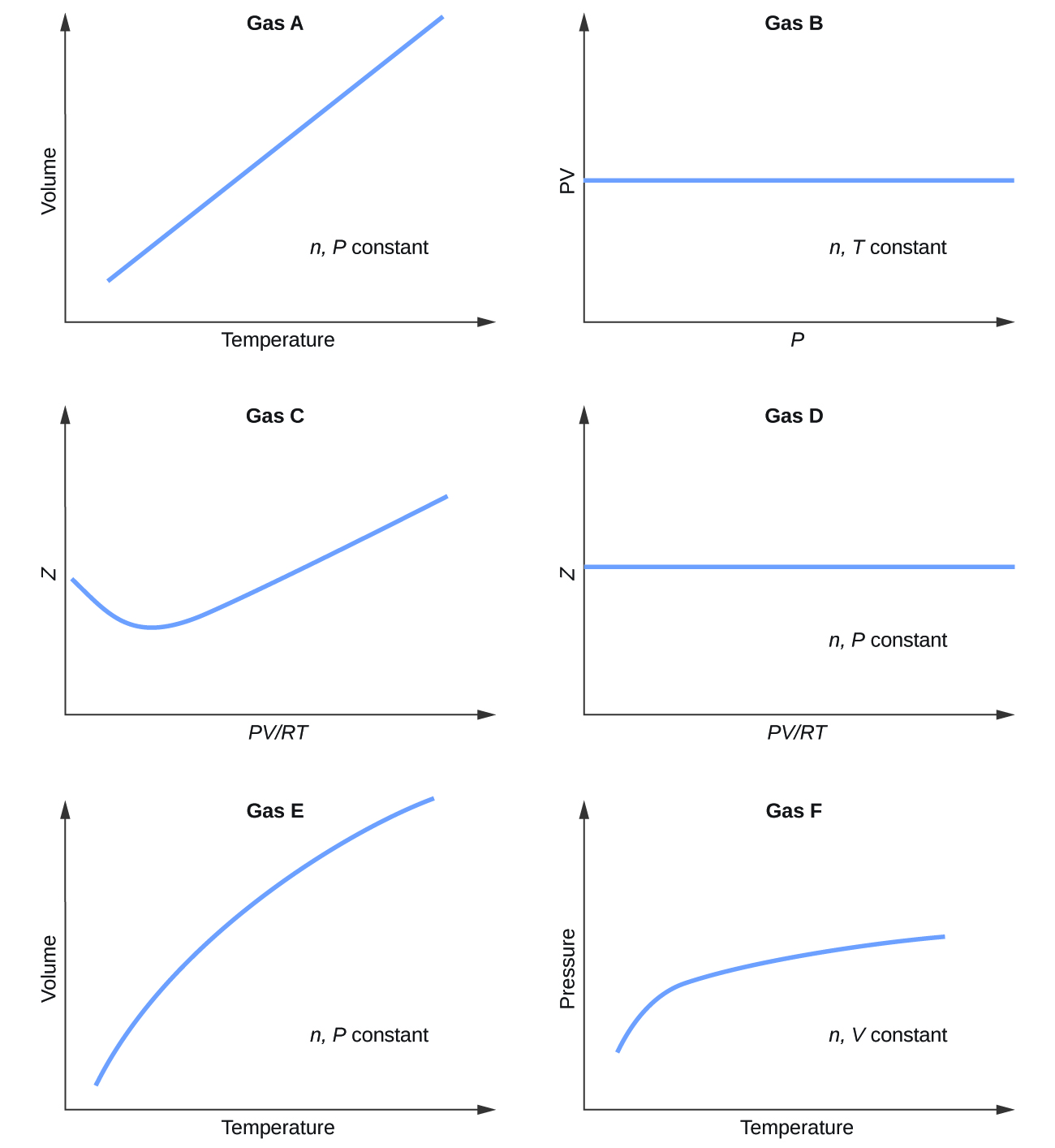
Non-Ideal Gas Behavior – Chemistry

What is the main difference between Process Plant Piping Engineering and Pipeline Engineering.?
Under what conditions do you expect a real gas such as hydrogen gas to behave like an ideal gas? - Quora

physical chemistry - Why do some gases have lower value of Z for a particular pressure? - Chemistry Stack Exchange
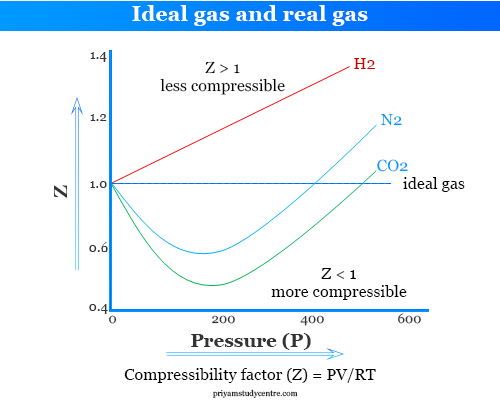
Ideal and Real Gases - Definition, Comparison, Properties
from
per adult (price varies by group size)