Establishing expiry date for clinical diagnostic reagents
By A Mystery Man Writer
Description
Product shelf life is an essential product performance requirement that, along with other design requirements, is used to determine the safety and efficacy of a clinical diagnostic
e1.jpg)
ASTM D4337-89(1995)e1 - Standard Test Methods for Analysis of

UNE EN ISO 23640:2015 In Vitro Diagnostic Medical Devices, 41% OFF
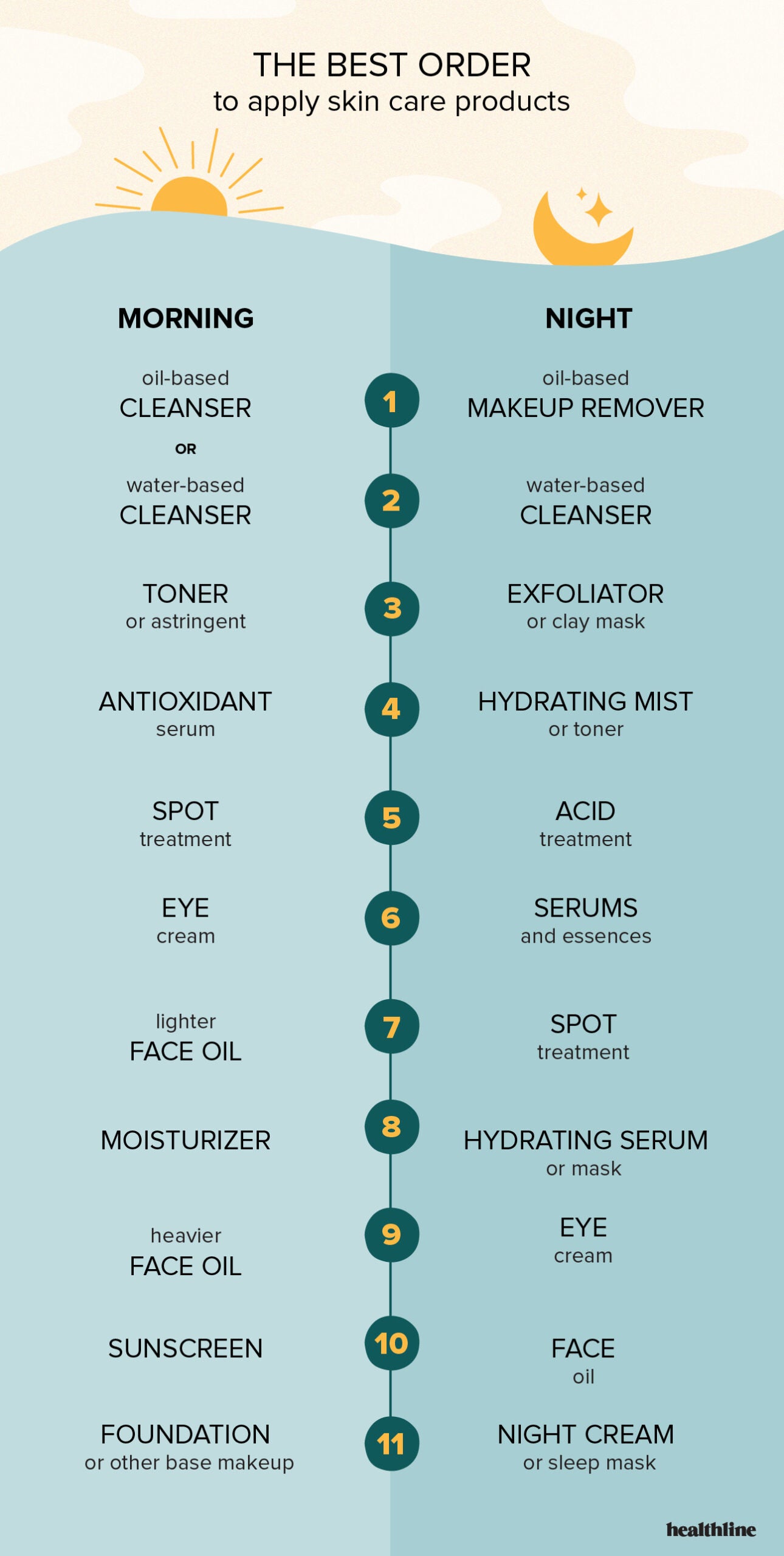
Skin Care Routine: What Is the Correct Order?

UNE EN ISO 23640:2015 In Vitro Diagnostic Medical Devices, 41% OFF

What Is Product Development? 7 Steps to Making a Product (2024

EP26Ed2IGE User Evaluation of Acceptability of a Reagent Lot
Focal Infection and Its Relation to Toxaemia of Pregnancy with or
National Action Plan to Build Australia's Diagnostic Technology
:max_bytes(150000):strip_icc()/hlt-tier-3-primary-best-fiber-supplements-ahuang-038-6844316380c64193a29fe2c3b79f2442.jpeg)
The 7 Best Fiber Supplements of 2024, According to a Dietitian

How to find out your eyesight prescription: Where and what to know

UNE EN ISO 23640:2015 In Vitro Diagnostic Medical Devices, 41% OFF
from
per adult (price varies by group size)







