SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1
By A Mystery Man Writer
Description
VIDEO ANSWER: Hello students: let's look at the question: l n, that integrate integration and 0 z minus 1 bracket, close d p by p here. Minus 1 is equal to minus 8.50 into 10 to the power minus 3 p by p, not plus 3.50 into 10. To the power minus 9. P
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

300 Solved Problems in Geotechnical Engineering
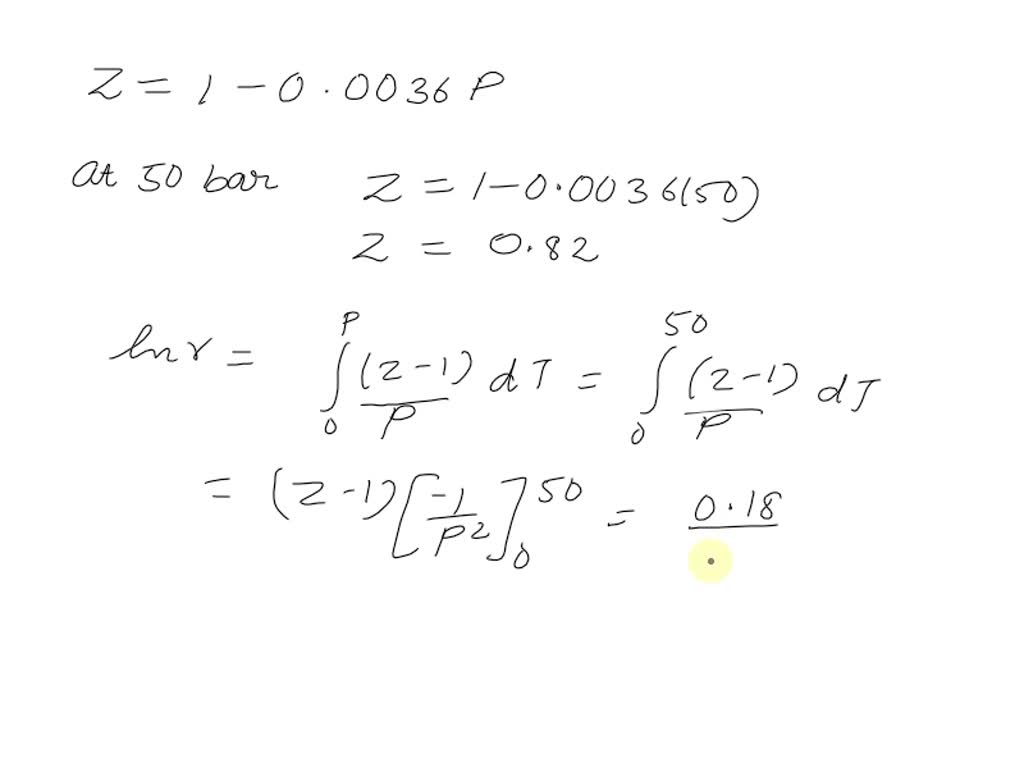
SOLVED: PROBLEM 3: The compressibility factor (Z) for nitrogen was measured at -100 °C from 10 to 50 bar pressure. The results are shown below: Pressure (bar) Compressibility factor (Z) 0 14

Soil water diffusivity and water content distribution during outflow experiment

Thermodynamics - 3-7 Ideal Gas Equation with compressibility factor

Computational Physics

Solved For a gas at a given temperature, the compression

EOS Residuales Thermodynamics & Chemicals Kinetics (ChBC-34), PDF, Heat

Che 4 Book, PDF, International System Of Units

Evaluation de la porosité de drainage à partir de limnigrammes de nappes

EXAMPLE PROBLEMS AND SOLUTIONS McGraw-Hill Education - Access Engineering

Solved NAME: 1.(a) Plot compression factor Z verses pressure

chapter 1 naval architecture architecture for the salvage engineer

Computation of The Compression Factor An, PDF, Gases

PDF) Solucionario de Física II Para Ciencias e Ingeniería, 7a ed

Physical Chemistry The Compression Factor (Z) [w/1 example]
from
per adult (price varies by group size)







