Solved An ideal gas initially at Pi, V;, and T; is taken
By A Mystery Man Writer
Description
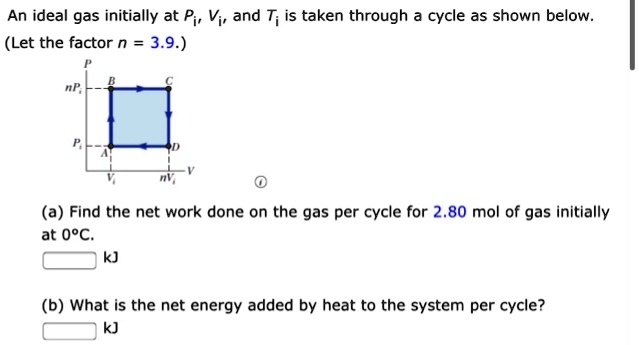
SOLVED: An ideal gas initially at Pi, Vi, and T; is taken through cycle as shown below (Let the factor n = 3.9.) (a) Find the net work done on the gas

1 mole of an ideal gas undergoes reversible isothermal expansion from an initial volume V_{1} to a final volume 10V_{1} and does 10 KJ of work. The initial pressure was 1times 10^{7}PaCalculate V_{1}
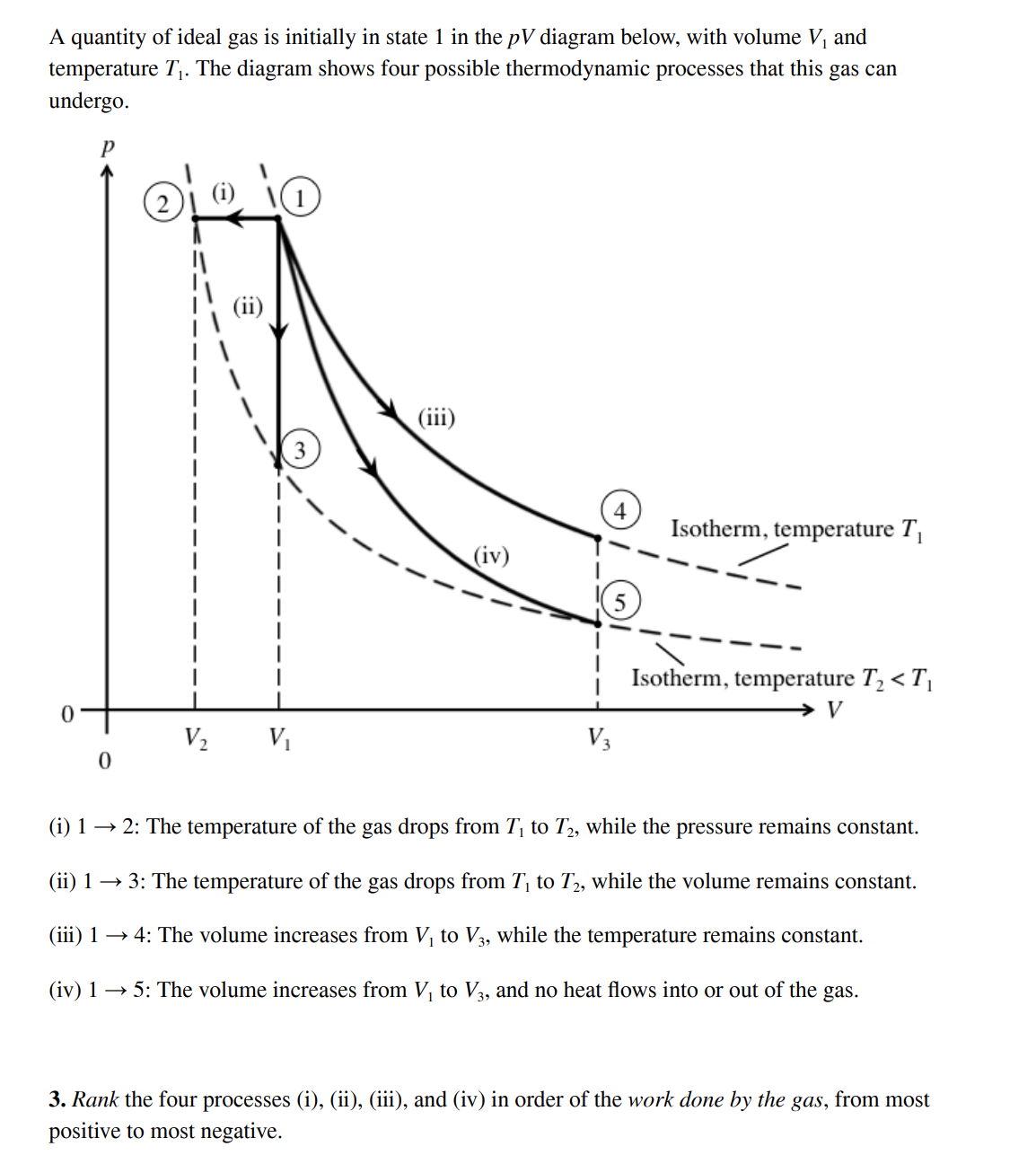
Solved A quantity of ideal gas is initially in state 1 in
1st law

A 4.00-L sample of a diatomic ideal gas with specific heat ratio 1.40, confined to a cylinder, is carried through a closed cycle. The gas is initially 1.00 atm and 300 K.
entropy

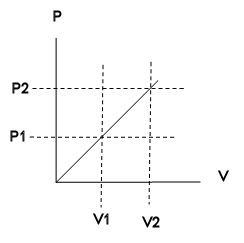
An ideal gas is initially at P1,V1 is expands to P2,V2 isothermally an

The state of an ideal gas is changed through an isothermal process at temperature T 0 as shown in figure. The work done by the gas going from state B to C

An ideal gas initially at P_i, V_i and T_i is taken through a cycle as shown below. Let the factor n = 3.7. a. Find the net work done on the gas
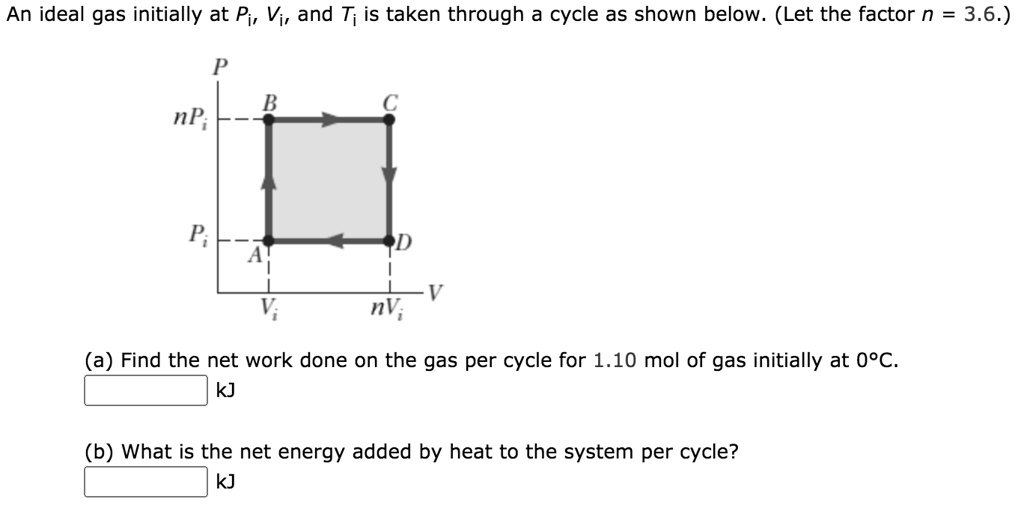
SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done

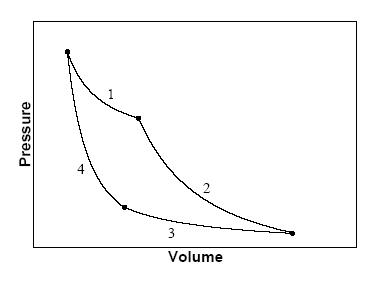
An ideal gas follows a process described by `PV^(2)=C` from `(P_(1), V_(1),T_(1)) to (P_

OpenStax College Physics, Chapter 13, Problem 22 (Problems & Exercises)
from
per adult (price varies by group size)





/i.s3.glbimg.com/v1/AUTH_59edd422c0c84a879bd37670ae4f538a/internal_photos/bs/2020/4/q/fwMAbzScegqDIfYyYkHw/2020-05-19t184957z-461916947-rc2urg968kbj-rtrmadp-3-health-coronavirus-usa.jpg)
