Aβ(1-42) tetramer and octamer structures reveal edge conductivity
By A Mystery Man Writer
Description

Structure of AqpZ tetramer and location of mutations. The

The amyloid-inhibiting NCAM-PrP peptide targets Aβ peptide
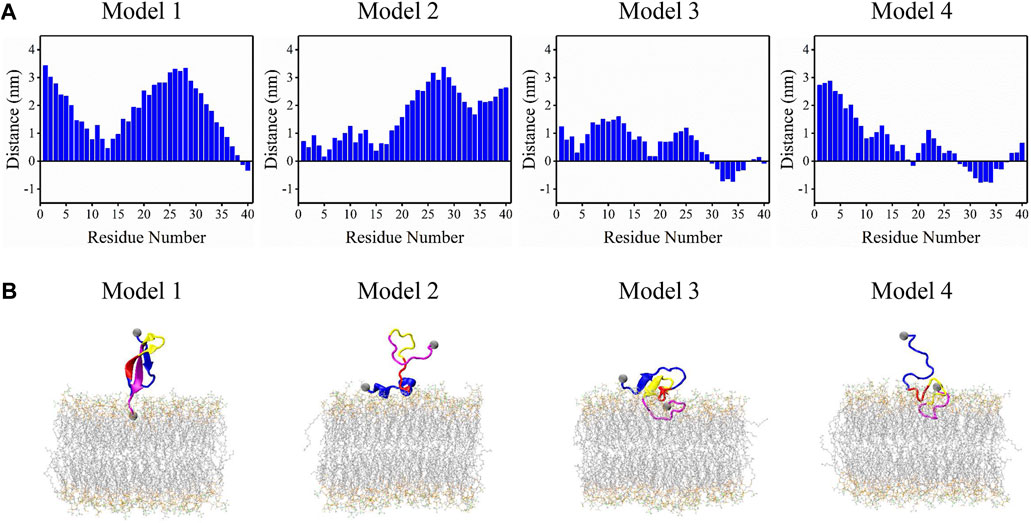
Frontiers Binding mechanism of full-length Aβ40 peptide to a mixed lipid bilayer

Eduard PUIG GOMÀ-CAMPS, Postdoctoral Associate

PDF) Aβ(1-42) tetramer and octamer structures reveal edge pores as a mechanism for membrane damage

Amyloid-Beta's Atomic Structure Reveals Alzheimer's Disease Toxicity Mechanism
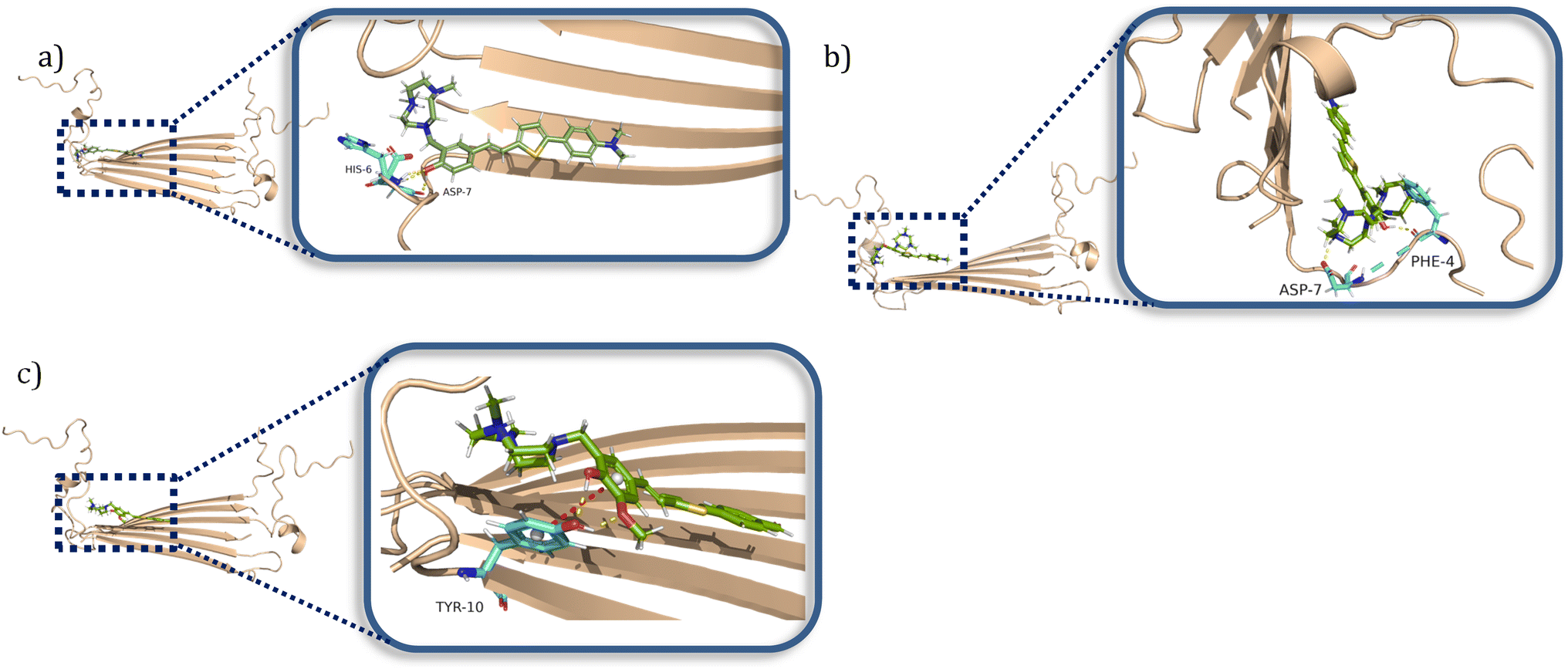
Amphiphilic stilbene derivatives attenuate the neurotoxicity of soluble Aβ 42 oligomers by controlling their interactions with cell membranes - Chemical Science (RSC Publishing) DOI:10.1039/D2SC02654F

Molecular dynamics simulations reveal the importance of amyloid-beta oligomer β-sheet edge conformations in membrane permeabilization - Journal of Biological Chemistry
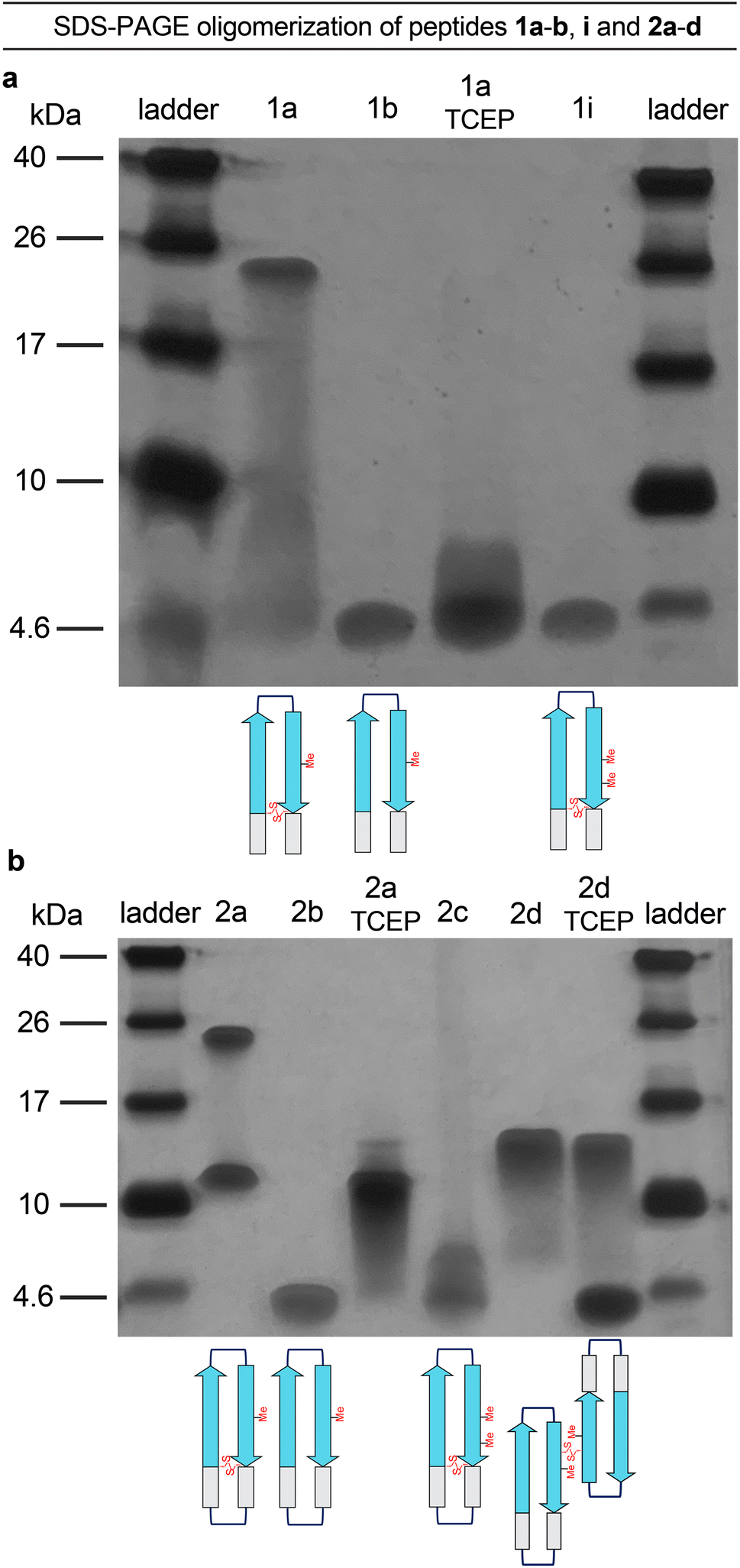
A β-barrel-like tetramer formed by a β-hairpin derived from Aβ - Chemical Science (RSC Publishing) DOI:10.1039/D3SC05185D
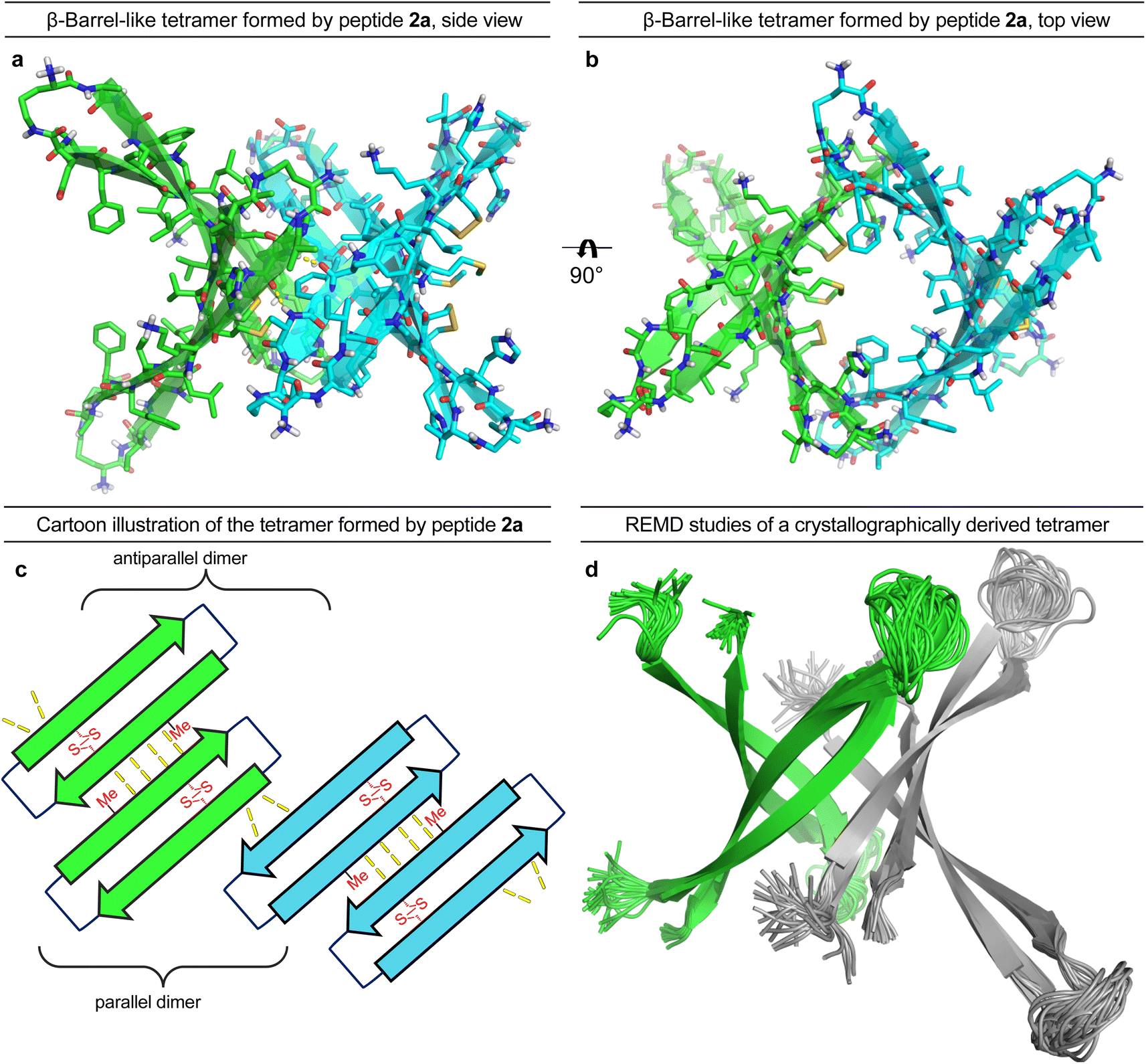
A β-barrel-like tetramer formed by a β-hairpin derived from Aβ - Chemical Science (RSC Publishing) DOI:10.1039/D3SC05185D

Exploring amyloid oligomers with peptide model systems - ScienceDirect
Structure of amyloid β25–35 in lipid environment and cholesterol-dependent membrane pore formation

RCSB PDB - 6RHY: Structure of pore-forming amyloid-beta tetramers

Carulla Laβ (@carullalab) / X
from
per adult (price varies by group size)





