The vapour pressure of a solution having 2.0 g of solute X (gram
By A Mystery Man Writer
Description
The vapour pressure of a solution having 2.0 g of solute X (gram atomic mass=32 g/mol) in 100 g of CS2 (vapour pressure =854torr) is 848.9 torr.The molecular formula of solute 1) X 2)X2 3)X4 4)X8
The vapour pressure of a solution having 2-0 g of solute X -gram atomic mass-32 g-mol- in 100 g of CS2 -vapour pressure -854torr- is 848-9 torr-The molecular formula of solute 1- X 2-X2 3-X4 4-X8
The vapour pressure of a solution having 2-0 g of solute X -gram atomic mass-32 g-mol- in 100 g of CS2 -vapour pressure -854torr- is 848-9 torr-The molecular formula of solute 1- X 2-X2 3-X4 4-X8

Colligative Properties: Definition, Types, and Examples

3 Ways to Calculate Vapor Pressure - wikiHow
A solution contains 500 g of LiCl (MM=42g/mol) in 1.5 L of water. What is the vapor pressure of water (in torr) over the solution at 25.0 oC? (The vapor pressure of

SOLVED: The vapour pressure of a solution having 2.0 g of solute X

Consider a binary mixture of volatile liquides. If at `X_(A)=0.4`, the vapour pressure of solution i

Culligative Properties, Abnormality in Molar Mass) The vapour pressure of a solution having 2.0 g of solute X (gram atomic mass = 32 g mol-) in 100 g of CS, (vapour pressure =

Molar Mass from Osmotic Pressure

Vapour pressure of a pure liquid X is 2 atm at 300 K. It is lowered to 1 atm on dissolving 1 g of Y in 20 g of liquid X. If
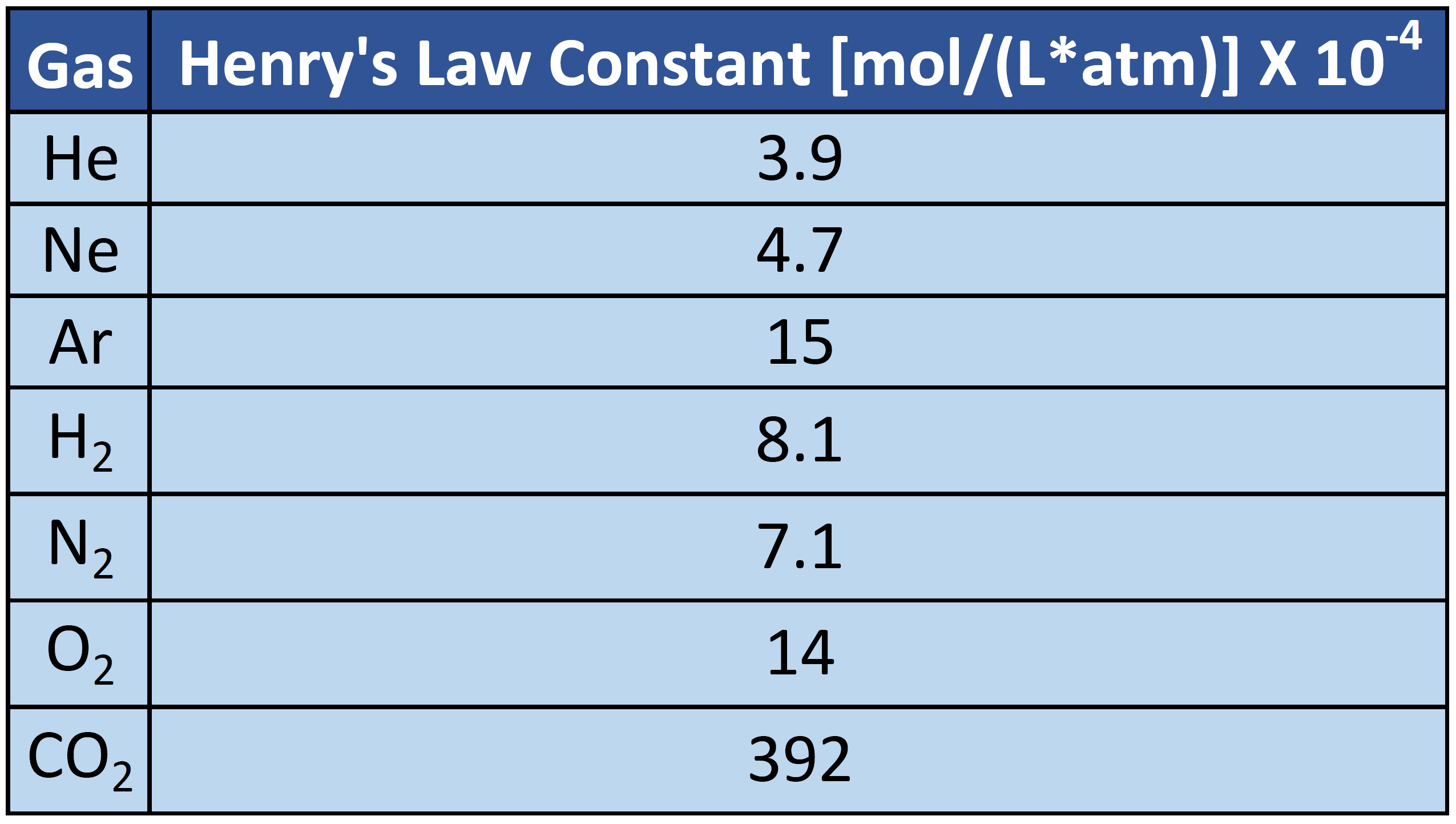
CH104: Chapter 7 - Solutions - Chemistry

How to Find Mol From Grams

Understanding Liquid Solutions: Methods of Expressing Concentration, Vapour Pressure Concepts, and Raoult's Law, PDF, Solution

SOLVED: The vapour pressure of a solution having 2.0 g of solute X

In ideal solution of non volatile solute B in solvent A in 2 : 5 molar ratio has vapour pressure 250

Solved 1. Assuming Raoult's Law applies, calculate the vapor
from
per adult (price varies by group size)







