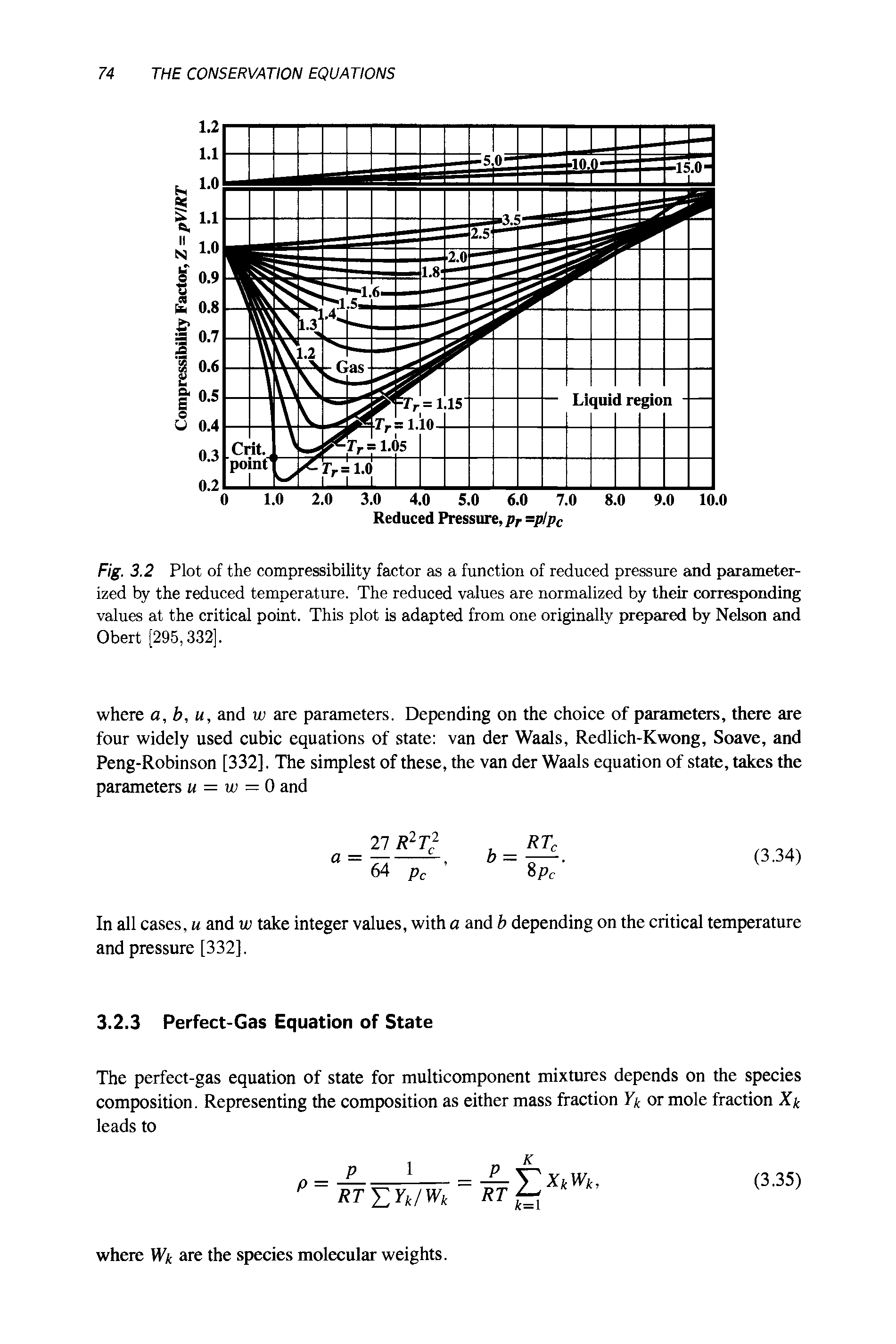
The role of the compressibility factor Z in describing the
By A Mystery Man Writer
Description
In this post I will give a recapitulation of the role the compressibilty factor Z plays in the volumetric behavior of gases. The purpose of this post is also to give some background to the first post of June , 2013 describing a compact, explicit equation for the vapor compressibility factor Z in the sub-critical…
Compressibility Factors Using the Soave-Redlich-Kwong Equation of State - Wolfram Demonstrations Project

The given graph represents the variation of compressibility factor Z vs P for three gases A, B and C.Identify the incorrect statements.

Objectives_template

Chemistry Desk: Effect of Pressure
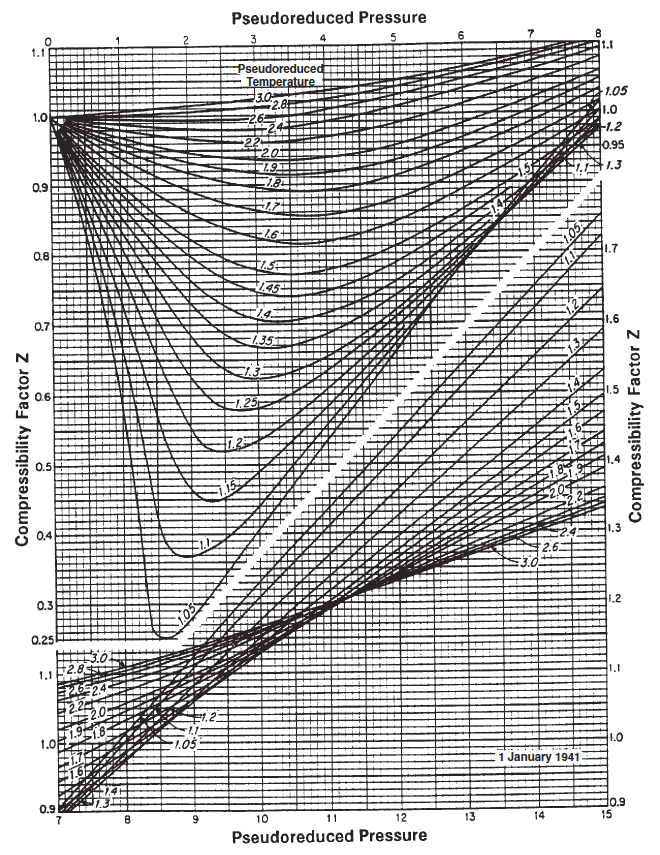
Real Gas Law - whitson wiki

Equation of state (excess compressibility factor Z À1 ¼ PV/(NkT) À 1 as

Compressibility factor (z): real gases deviate from ideal behav-Turito

Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt

ars.els-cdn.com/content/image/3-s2.0-B978012809597

Compressibility Factor - an overview

Non-Ideal Gas Behavior Chemistry: Atoms First
from
per adult (price varies by group size)