Solved RT B 2. The compressiblity factor for a gas is
By A Mystery Man Writer
Description
Answer to Solved RT B 2. The compressiblity factor for a gas is

1st law

The compressibility factor for a real gas at high pressure is :- (1) 1-pb/RT (2) 1+RT/pb (3) 1

Thermodynamics: An Engineering Approach - 5th Edition - Part II by 黑傑克 - Issuu
Solved] please help with this question There are regimes in which the
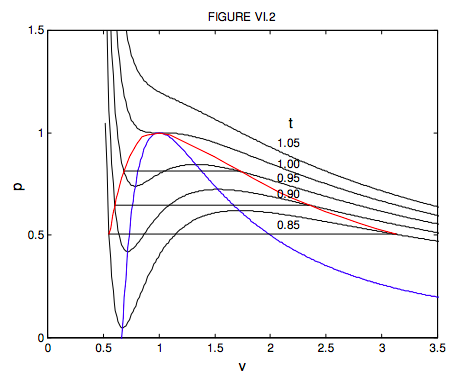
6.3: Van der Waals and Other Gases - Physics LibreTexts

What is the compressibility factor (Z) for 0.02 mole of a van der Waals's gas at pressure of 0

Real Gases Introductory Chemistry
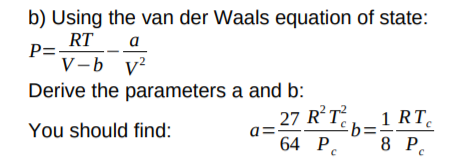
Answered: b) Using the van der Waals equation of…

Gaseous State

JEE: Van der Waals Equation, Chemistry By Unacademy

The compressibility factor for a real gas is expressed by, z =1+ BP / RT. The value of B at 500 K and 600 bar is 0.0169 L / mol. Find the

COMPRESSIBILITY FACTOR
from
per adult (price varies by group size)







