If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
By A Mystery Man Writer
Description

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

For one mole of a Van der Waals gas when b=0 and T=300 K, the PV vs. 1/V plot is shown below.

Multiple Choice Questions on Gas Laws and Kinetic Theory, PDF, Gases

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool

If Z is a compressibility factor, van der Waals' equation at low press

If Z is a compressibility factor, van der Waals' equation at low press
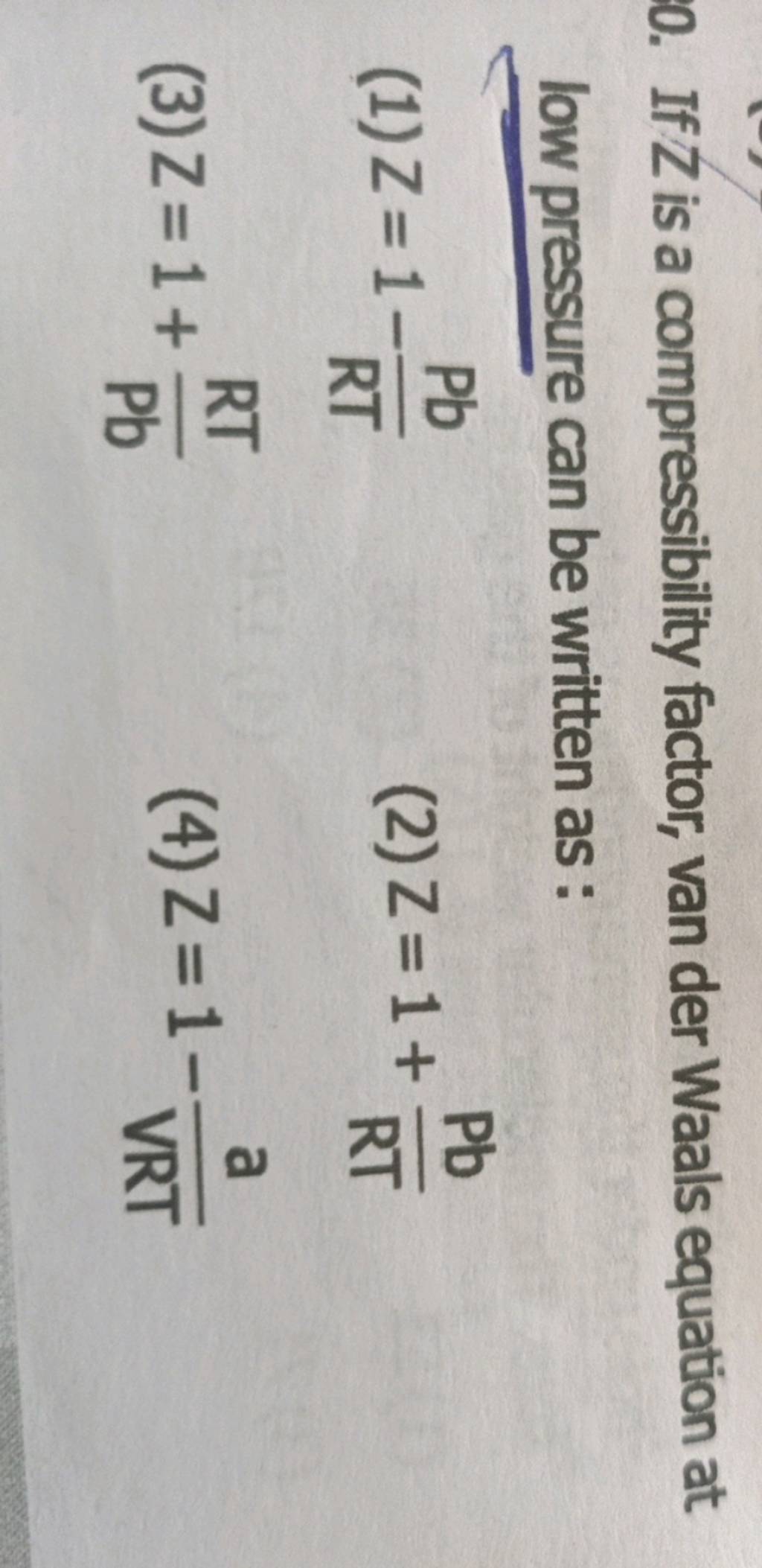
If Z is a compressibility factor, van der Waals equation at low pressure ..

012 IfZ is a compressibility factor, van der Waals equation low pressure can be written as: [2014] RT I-끔 (C) Z-I+ Z=1+ (B) Ζ=I.RT (D) Z=l- _ pb VRT

The compression factor (compressibility factor) for `1 mol` of a van der Waals gas at `0^()C` an

SOLVED: If PR=3, TR=2.0, the compressibility factor (Z) is equal to: (2 Points) a. Z=0.85 b. Z=0.55 c. Z=1.5 960=Zp
from
per adult (price varies by group size)