Ideal gas law, Definition, Formula, & Facts
By A Mystery Man Writer
Description
Ideal gas law, relation between the pressure P, volume V, and temperature T of a gas in the limit of low pressures and high temperatures, such that the molecules of the gas move almost independently of each other. In such a case, all gases obey an equation of state known as the ideal gas law: PV =

Ideal Gas Law Equation Compressibility Of Natural Gas - Chemistry

Avogadro's Law: Definition, Formula, Derivation, Examples

The ideal gas law (PV = nRT) Intermolecular forces and

Gay-Lussac's Law: Statement, Formula, Explanation, Example & FAQs

Calculating Pressure of an Ideal Gas - Derivation

Avogadro's Law - Definition, Formula, Examples
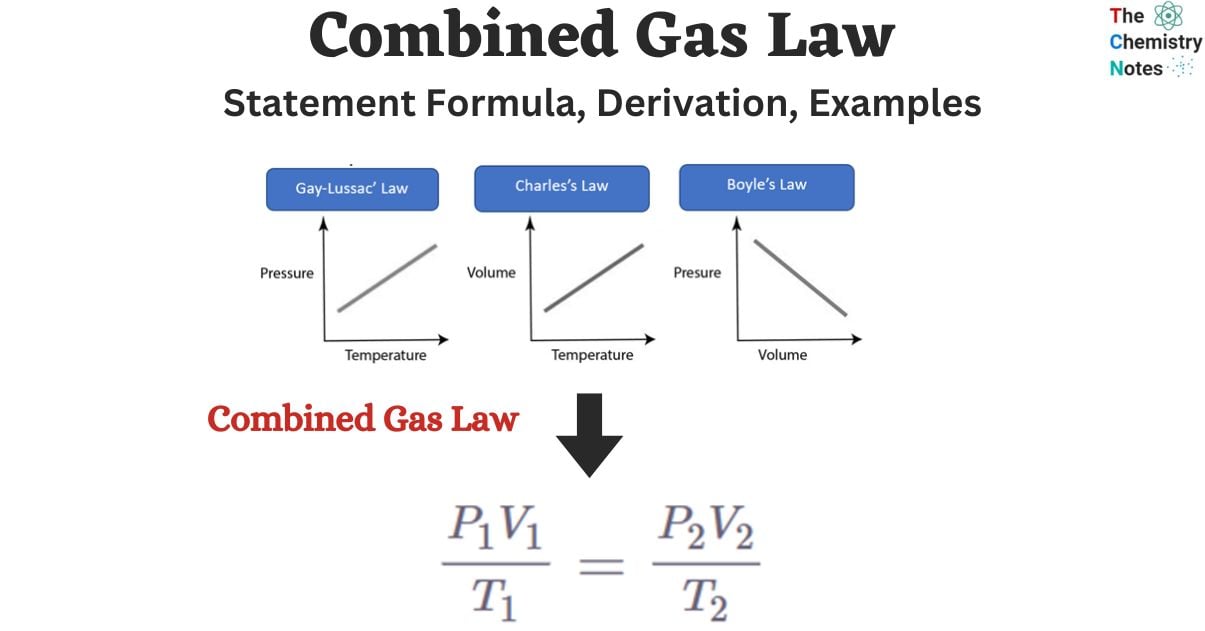
Combined Gas Law: Formula, Derivation, Examples
:max_bytes(150000):strip_icc()/143058853-56a12f375f9b58b7d0bcdc3c.jpg)
Combined Gas Law Definition and Examples

Ideal Gas Law — Overview & Calculations - Expii

Ideal Gas Constant (R) - Universal Gas Constant
from
per adult (price varies by group size)







