How to Calculate Normality of a Solution
By A Mystery Man Writer
Description
The normality of a solution is the gram equivalent weight of a solute per liter of solution. Here are examples of how to calculate the normality.
How to calculate the normality of a solution - Quora

Calculate normality of 2.1 Chemistry Questions
The number of ions present in 0.2 mole of sodium chloride is

11) 1000 (2) TUU 21. Normality of 10% (wV) H,SO, solution is nearly (1) 0.1 (2) 0.2 (3) 0.5 (4) 2

Normality,Molality,Molarity,Mole fraction,Formality

Normality Calculation - Chemistry
What is the normality of solution obtained by mixing 100 ml of 0.2 M H2SO4 with 100 ml of 0.2 M NaOH? - Quora

Normality Calculation - Chemistry
Calculate the normality of a solution containing 62.3 g of hydrated copper sulphate in 500ml of solution (Cu= 63, S= 32, O=16, H=1)

Calculate the normality of HCl solution whose 500 ml is utillised to neutralise the 1500 ml of

NormalityHow to calculate normality of a solution
Normality Definition & Formula , Formality Formula, Solved Examples
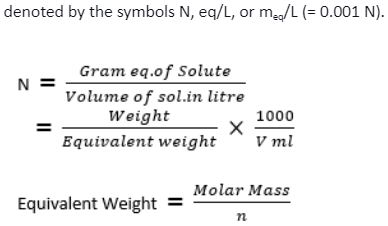
Normality and its formula - Everything you need to know
What is the molarity of 0.4 normality of H2SO4 solution? - Quora
Lesson 7: Acids and Bases
from
per adult (price varies by group size)