Applications for Medical Device Investigational Testing Authorizations Guidance Document
By A Mystery Man Writer
Description
Applications for Medical Device Investigational Testing Authorizations Guidance Document

validation and verification of medical device.pptx

Overview of Medical Device Clinical Trials - ScienceDirect

/wp-content/uploads/120650
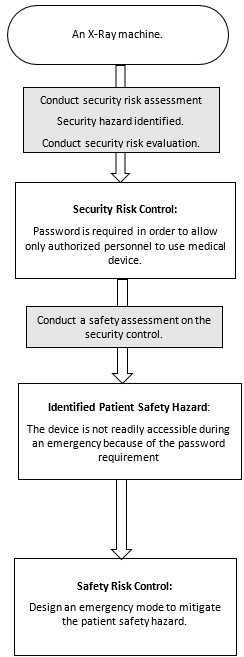
Guidance Document: Pre-market Requirements for Medical Device
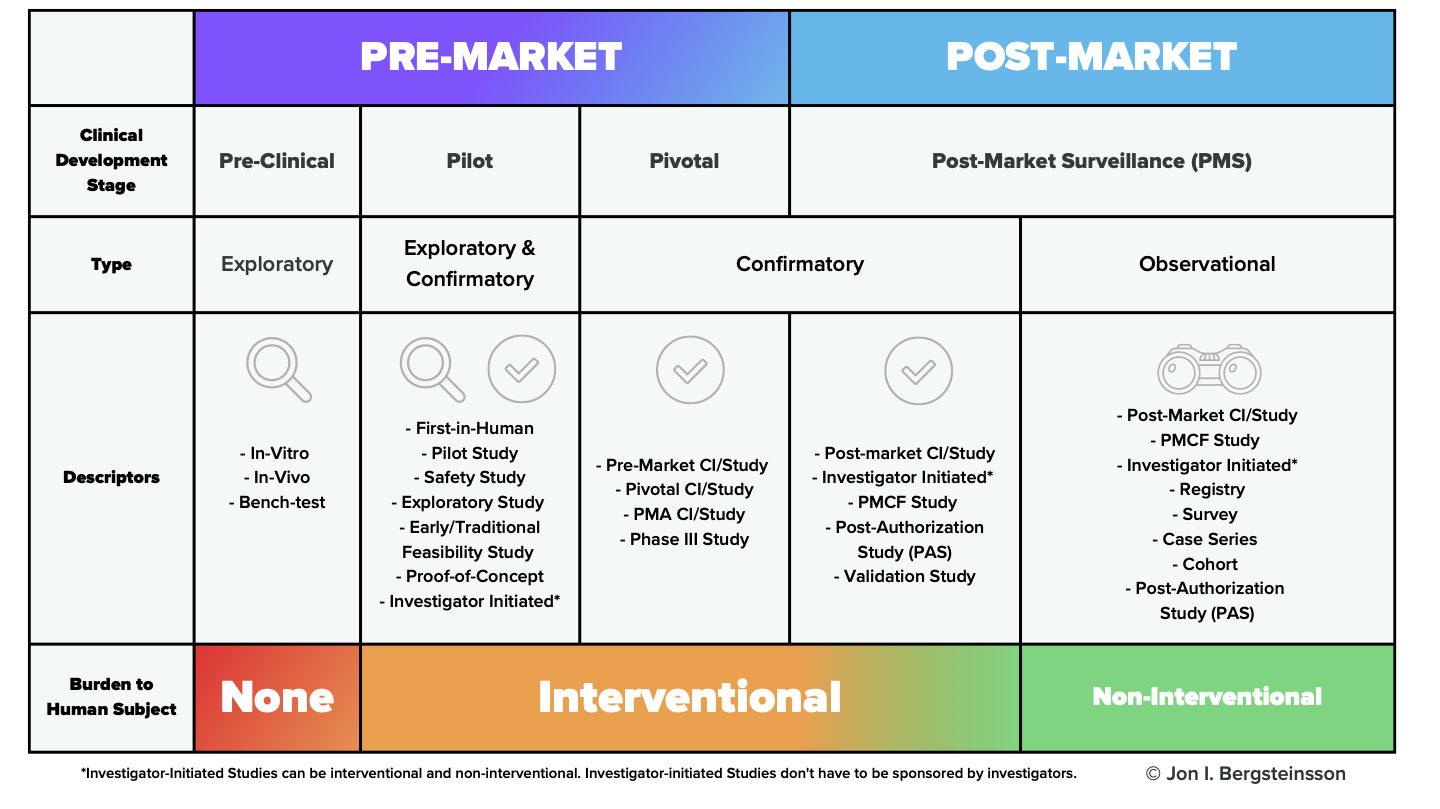
ISO 14155:2020 Compliant Clinical Investigations - The Ultimate Guide

Regulatory oversight of genetic testing in Canada: Health Canada
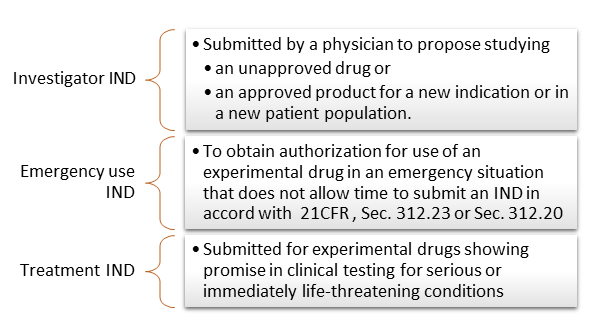
Investigational New Drug (IND) Application

Regulatory oversight of genetic testing in Canada: Health Canada perspective

/wp-content/uploads/Pictur
from
per adult (price varies by group size)