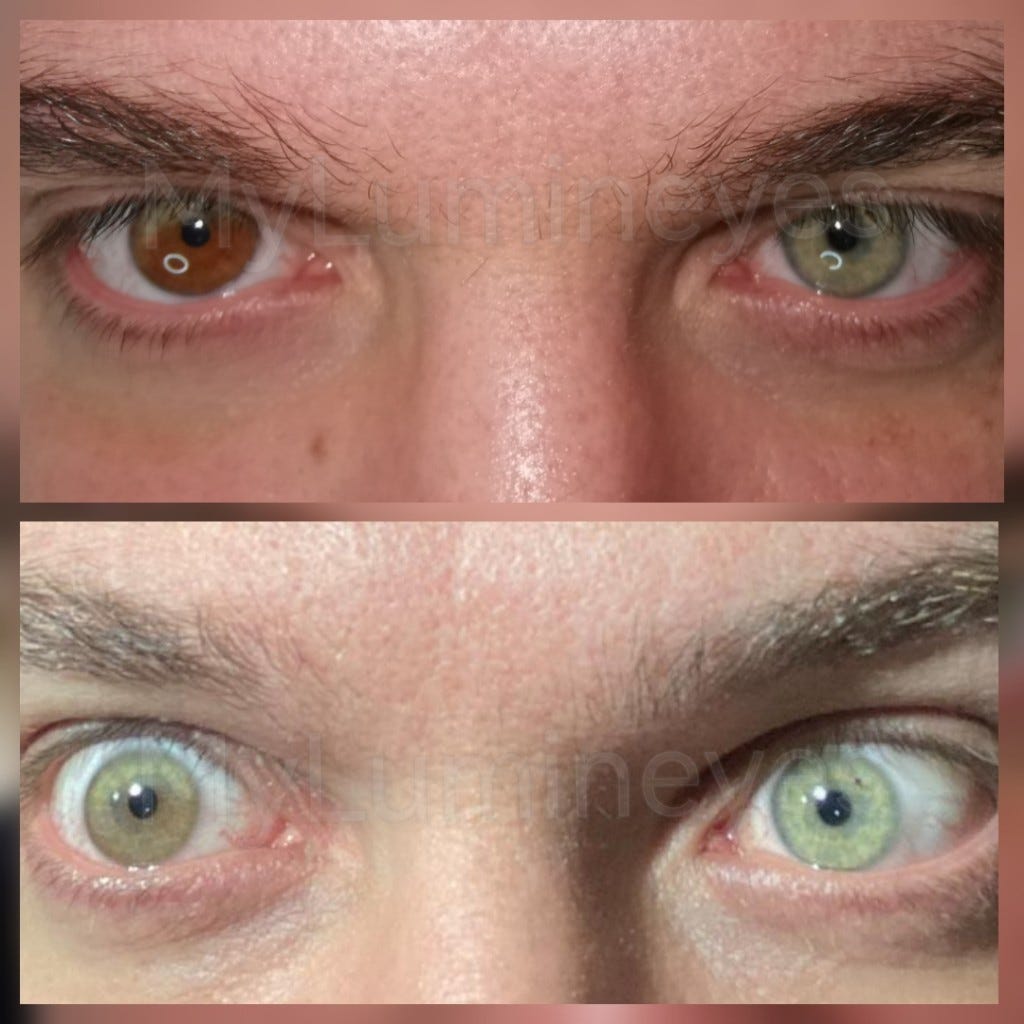
Color change is only device modification. Is a new 510k required? - Medical Device Academy
By A Mystery Man Writer
Description
This article explains the process for determining if a color change and other material changes require a new 510k prior to implementing the change.
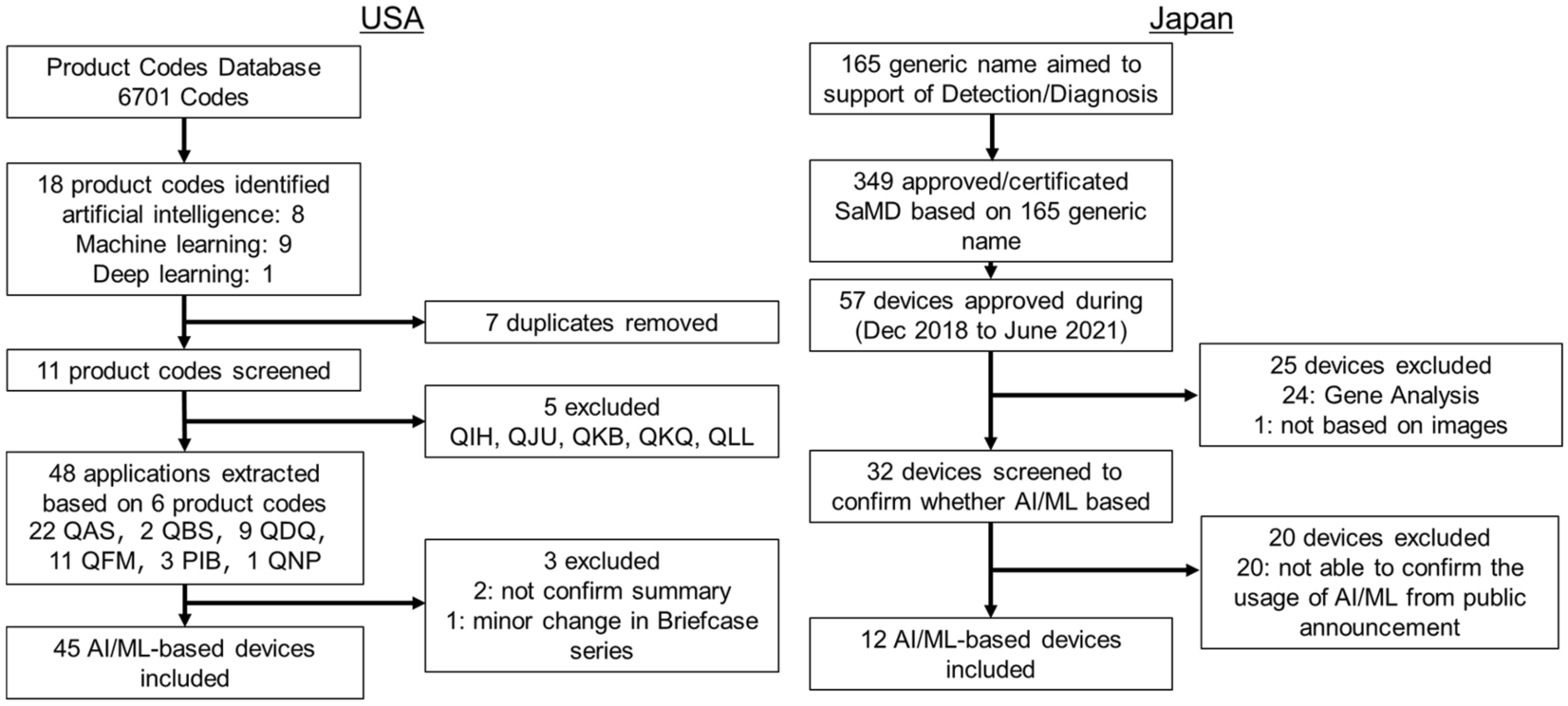
Systematic analysis of the test design and performance of AI/ML

Usability Testing of Medical Devices, FDA Guidance
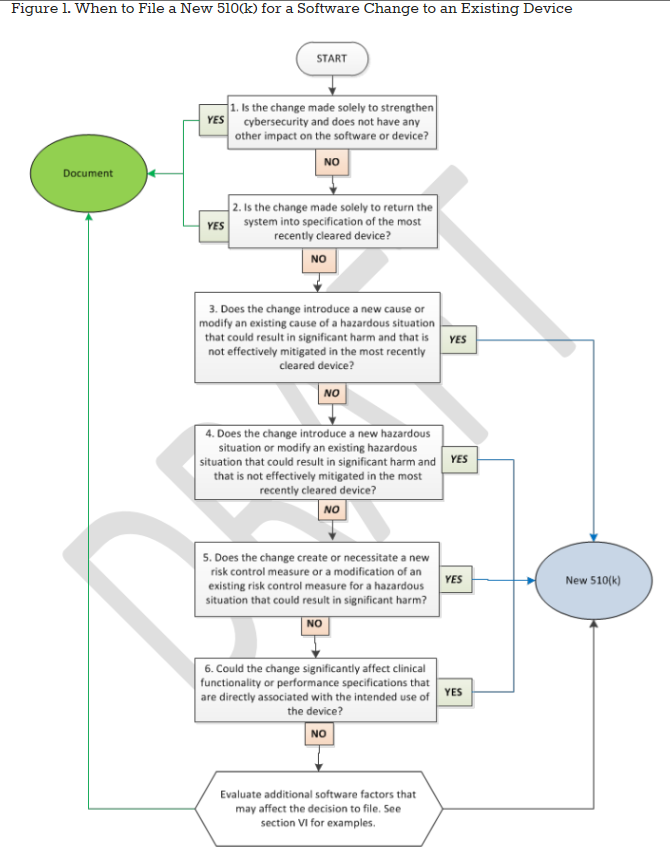
FDA Issues Proposed Guidance for Changes to Medical Device

Medical Device Regulations - 510(k) Process

FDA Guidance 2023 on ISO 10993-1: What Changed?

Schematic representation of medical device premarket review
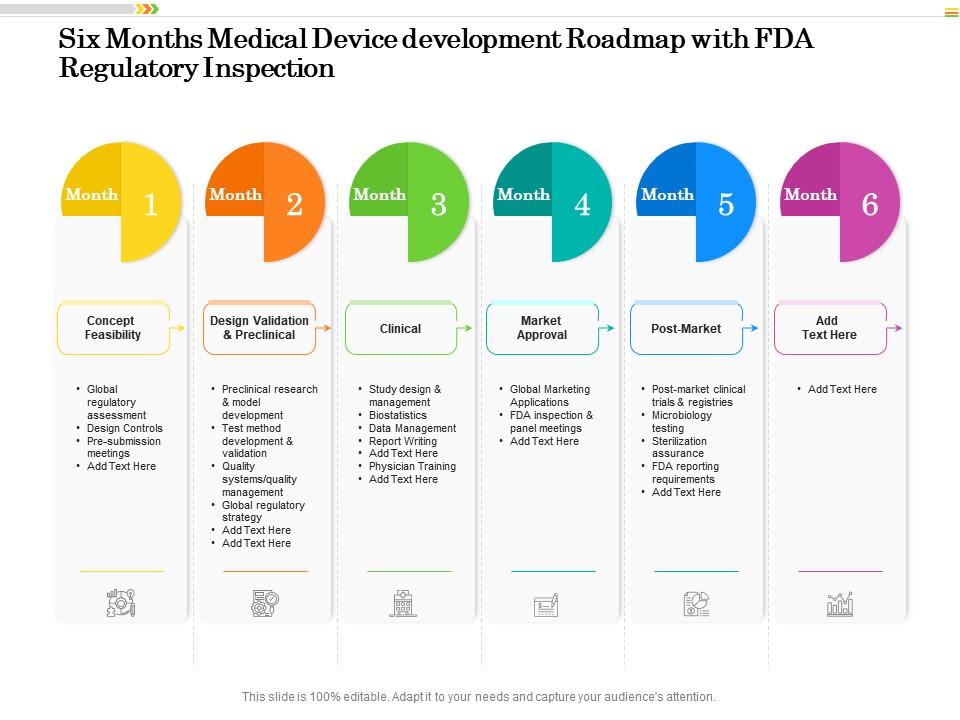
Six Months Medical Device Development Roadmap With FDA Regulatory Inspection, PowerPoint Slides Diagrams, Themes for PPT
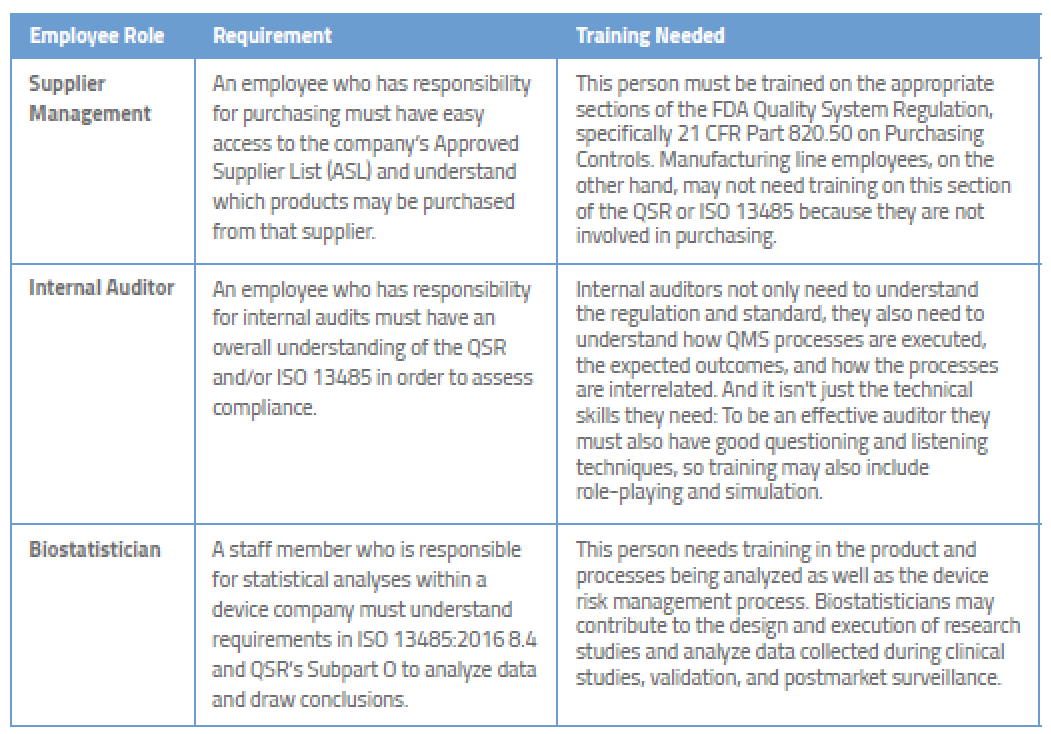
Medical Device Regulatory Training Requirements for Employees
Understanding the New FDA Guidance on Changes to a 510(k)

Labels for Medical Devices
FDA
The FDA 510(k) Process: Setting the Stage for a Successful

FDA
from
per adult (price varies by group size)