42g of N₂ react with excess of O₂ to produce NO. Amount of NO formed is a.60g b.32g c.45g d.90g
By A Mystery Man Writer
Description
Share your videos with friends, family and the world

Solved Nitrogen gas can be combined with oxygen to form

PDF) Answers Power Cell

Solutions Dinesh, PDF, Molar Concentration
42. If 25 g N2 reacts with H2 then amount of NH3 produced in


Mole Concept PDF, PDF, Mole (Unit)
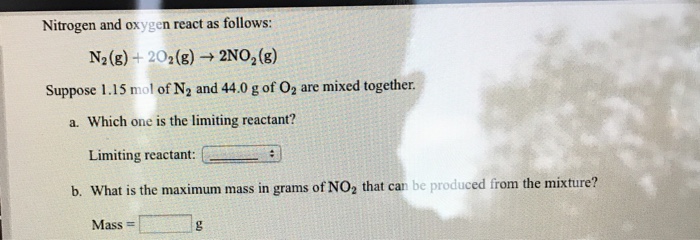
Solved Nitrogen and oxygen react as follows: N2(g)-202(g

Mole Concept PDF, PDF, Mole (Unit)
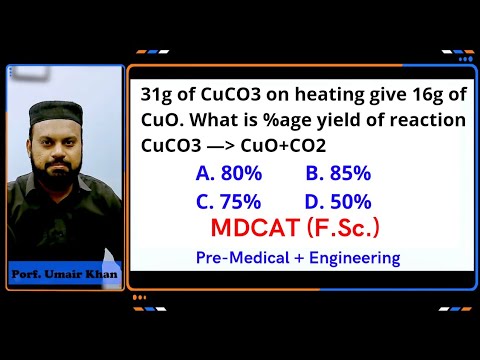
31g of CuCO3 on heating give 16g of CuO. what is %age yield of

31g of CuCO3 on heating give 16g of CuO. what is %age yield of reaction. 80% 85% 75% 50%

Limiting Reagent What happens in a chemical reaction, if there is

According to the following reaction, how many grams of oxygen gas are required for the complete reaction of
from
per adult (price varies by group size)







