For H(2) gas, the compressibility factor,Z = PV //n RT is
By A Mystery Man Writer
Description
For H(2) gas, the compressibility factor,Z = PV //n RT is

Non-Ideal Gas Behavior Chemistry: Atoms First
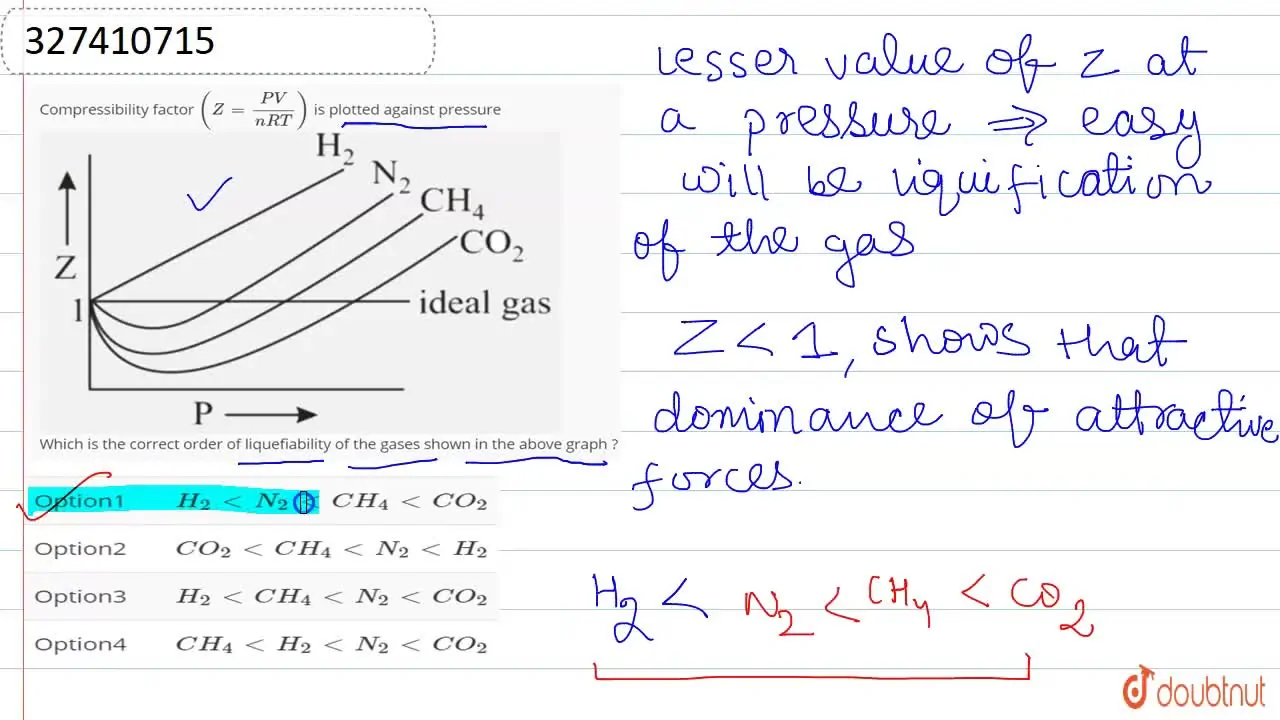
Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Deviations from ideal gas behaviour, intermolecular forces, Van der Waals equation of state, compressibility factors and the critical pressure and critical temperature of a gas revision notes doc brown's chemistry UK advanced

34. What is Compressibility factor? [Imp.Q] A: The ratio of the actual m..
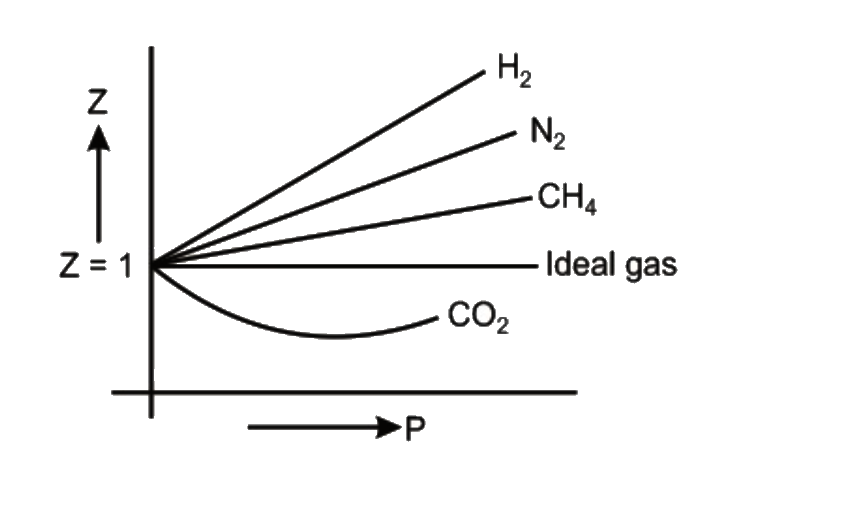
Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Gas Compressibility - an overview

Why H2 gas does not show the Joule-Thomson effect?

1. The compressibility factor, z, is the ratio of

34. What is Compressibility factor? [Imp.Q] A: The ratio of the actual m..
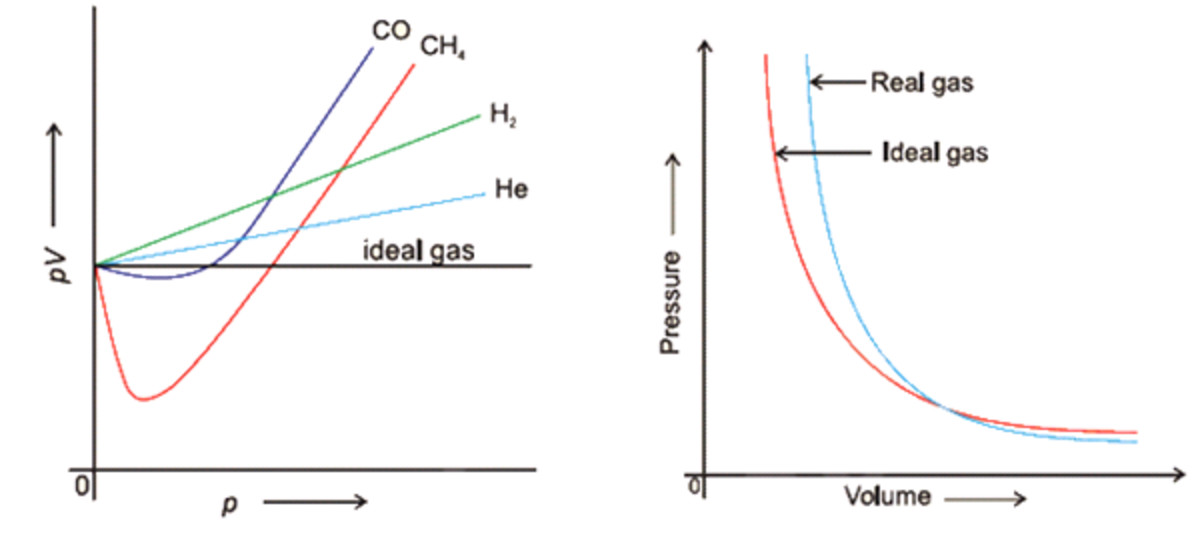
Real Gases - Chemistry, Class 11, States of Matter

The given graph represent the variations of Z Compressibility factor Z PV nRT versus p for three real gases A B and C Identify the only incorrect statement

For an ideal gas, the value of compressibility factor `Z(=(pVm)/(RT))` is
from
per adult (price varies by group size)







